International Surgery Case Reports
OPEN ACCESS | Volume 8 - Issue 1 - 2026
ISSN No: 2836-2845 | Journal DOI: 10.61148/2836-2845/ISCR
Bratko Aikaterini1, Doumas Michael2, Zacharioudakis Georgios1, Mpallas Konstantinos1, Tzortzi Iliana3, Marouda Christina4, Savvas Ioannis3, Kamparoudis Apostolos1*
1Fifth Department of Surgery, Aristotle University of Thessaloniki, Thessaloniki, “Hippokration” General Hospital of Thessaloniki, Greece.
2Second Propedeutic Department of Internal Medicine, Aristotle University of Thessaloniki, “Hippokration” General Hospital of Thessaloniki, Greece.
3Companion Animal Clinic, Faculty of Veterinary Medicine, School of Health Sciences, Aristotle University of Thessaloniki, Thessaloniki, Greece.
4Laboratory of Pathology, School of Veterinary Medicine, Faculty of Health Sciences, Aristotle University of Thessaloniki, Greece.
*Corresponding author: Kamparoudis Apostolos, Fifth Department of Surgery, Aristotle University of Thessaloniki, Thessaloniki, “Hippokration” General Hospital of Thessaloniki, Greece.
Received: February 02, 2026 | Accepted: February 20, 2026 | Published: February 26, 2026
Citation: Aikaterini B, Michael D, Georgios Z, Konstantinos M, Iliana T, Christina M, Ioannis K, Apostolos K. (2026) “Renal Sympathetic Denervation is not Harmful in Sepsis-Induce Shock. An Experimental Porcine Model.”, International Surgery Case Reports, 8(1); DOI: 10.61148/2836-2845/ISCR/114.
Copyright: © 2026. Kamparoudis Apostolos. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Background: Septic shock is a life-threatening condition characterized by systemic inflammation, profound hemodynamic instability, and multi-organ dysfunction, with acute renal failure being a major contributor to mortality. Sympathetic nervous system (SNS) hyperactivation plays a key role in septic shock pathophysiology. Renal sympathetic denervation (RSD), an interventional approach used for treatment of resistant hypertension, may modulate sympathetic dysregulation; however, its effects during septic shock remain insufficiently studied.
Methods: Fourteen Large White pigs were randomized into two groups: bilateral renal sympathetic denervation (R group, n=9) and control (C group, n=5). Following surgical intervention, fecal peritonitis was induced to establish hyperdynamic septic shock. Hemodynamic parameters, urine output, laboratory biomarkers (inflammatory, coagulation, renal, hepatic, and electrolyte indices), and histopathological changes in major organs were assessed preoperatively, postoperatively, and 6 hours postoperatively of shock induction.
Results: RSD caused an immediate and sustained reduction in systolic blood pressure without significant changes in diastolic blood pressure, heart rate and urine output compared with controls. During septic shock, RSD was associated with significantly greater prolongation of prothrombin time and international normalized ratio, indicating enhanced coagulation system disturbance. Renal function deterioration, inflammatory response (WBC, CRP), hepatic enzymes, and electrolyte imbalances progressed similarly in both groups, with no statistically significant intergroup differences. Histopathological findings did not differ significantly between groups across examined organs.
Conclusions: In a porcine model of fecal peritonitis–induced septic shock, bilateral renal sympathetic denervation significantly alters hemodynamic stability and coagulation parameters but does not modify renal failure progression, inflammatory response, or hepatic dysfunction. These findings suggest that renal sympathetic activity influences cardiovascular and coagulation responses during septic shock without conferring any potential risk during septic shock.
Renal sympathetic denervation, Sepsis, Septic shock, Organ failure, inflammatory response
Septic shock remains a life-threatening condition characterized by widespread systemic inflammation and vascular dilatation, leading to multi-organ dysfunction. The complexity of this severe condition contributes to a high mortality rate in critically ill patients despite advancements in clinical care (1). According to the third international consensus definition (Sepsis-3), septic shock is defined as persistent hypotension with mean arterial pressure of less than 65 mmHg, requiring vasopressors to keep it above 65mmHg and elevated levels of serum lactate of more than 2 mmol/L and suspicion of infection (2). Peritonitis originating from abdominal infections, such as like bowel perforation constitutes one of the causes of sepsis and septic shock (3). The sympathetic nervous system (SNS)- with renal nerves detected circularly around renal arteries- plays a crucial role in septic shock pathophysiology because sympathetic hyperactivation modifies vascular stability and organ failure (4).
Renal sympathetic denervation (RSD) was introduced a couple of decates ago as a therapeutic option in conditions such as resistant hypertension and other cardiovascular disease (5, 6, 7, 8). Despite the initially controversial results, recent studies confirm effectiveness and safety of the method in controlling resistant hypertension (9, 10). However, by interrupting renal sympathetic nerve activity this intervention have detrimental effects in septic shock, due to the irreversible inhibition of the SNS.
Nevertheless, such severe conditions as septic shock generate obstacles to conducting studies on humans. For this reason experimental animal models are selected as means to get insight to humans. Pigs are very similar to human animals in terms of anatomy, physiology and genetics, with their immune effectors to be comparable to human structures and functions (11). Still there are very few studies in the international literature dealing with the impact of renal sympathetic denervation on systemic, organic and inflammatory, response in septic shock environment, but none of them was conducted in experimental porcine animal models (1, 12, 13, 14).
Therefore the present study was designed aiming to inspect whether renal sympathetic denervation affects systemic response into a hyperdynamic state of septic shock and, simultaneously, to determine its degree of influence either locally in kidneys or at a systemic level. This knowledge may contribute to clinical implications regarding application of RSD for treatment of arterial hypertension, influencing the indications and contraindications of the method.
Materials and Methods
Animals and preparation
The present study was approved by the Bioethical Committee of Aristotle University of Thessaloniki, the Ethics Committee of Companion Animal Clinic of School of Veterinary Medicine of Aristotle University of Thessaloniki (Registration num:EL-54-BIOexp-18) and Animal Care Committee of the Region of Central Macedonia of Greece (Protocol num: 54162(167)/09-02-2024)
The study included a total number of 14 Large White pigs, female and male, aged 3-4 months and body weight 25±5kg. All animals were transported to the Companion Animal Clinic of School of Veterinary Medicine of Aristotle University of Thessaloniki, 24 hours prior to the day of the experiment and were housed in the stable facility of the inpatient unit in specially designed stainless-steel cages for pigs, with free access to food and water and under controlled environmental conditions (temperature 18–22 °C, relative humidity 55–65%, and a 12-hour light/dark cycle).
Anesthesia and ventilation
All animals received pre-anaesthetic medication consisting of midazolam (0.1 mg/kg, Richmond Vet Pharma Inc, Buenos Aires, Argentinae), butorphanol (0.1 mg/kg, Vetviva Richter GmbH, Wels Austria), medetomidine (0.1 mg/kg, Eurovet Animal Health, Bladel, Nederlands), and ketamine (10 mg/kg, Vetviva Richter GmbH, Wels, Austria), administered intramuscularly into the psoas muscles. Then animals were subsequently transferred on the operating table of a fully equipped experimental operating theatre of the clinic.
Catheterization of the auricular vein was performed using an abbocath (22G, Polymed Medicure Limited, New Delhi, India)to establish peripheral venous access. General anesthesia was induced with intravenous administration of propofol (2-3 mg/kg, CP-Pharma Handlesgesellschaft mbH, Burgdorf, Germany) followed by endotracheal intubation with a size 7–8 endotracheal tube (Jiangsu Hendhong Medical technology Co. Ltd, Yizheng, China). General anesthesia was maintained with administration of 100% oxygen and Isoflurane (1-2%, Piramal Critical Care B.V. Inc, Voorshcoten, Nederlands) until the end of the study. Hemodynamic support was provided by intravenous administration of Lactated Ringer’s solution (10 mL/kg/h, Bradex Inc, Krioneri, Greece). In addition, intravenous ampicillin (5 g, Intervet Hellas Inc., Athens, Greece) was administered as preoperative antimicrobial prophylaxis (15).
Experimental protocol and Surgical Technique
All laboratory animals were randomly allocated into two groups:
Abdominal surgical field was prepared by using 10% povidone–iodine solution (Betadine, Lavipharm Inc, Athens, Greece) and applying sterile draping for sterilization. A midline laparotomy incision was performed in order to enter into the peritoneal cavity through the musculoaponeurotic layers of the abdominal wall.
Bladder catheterization was performed with placement of a suprapubic urinary catheter (14Fr 2way foley, Shangai International Holding Corp. GmbH, Hamburg, Germany). Subsequently, bilateral incision of the lateral peritoneal reflections was carried out to mobilize the colon away from the surgical field and to allow access to the retroperitoneal space, the kidneys, and the renal vessels.
In Group R, circumferential dissection and complete skeletonization of the renal arteries and their branches were performed bilaterally along their entire course, from their origin at the abdominal aorta to the renal hilum. Perivascular tissue was excised to ensure interruption of bilateral renal sympathetic neural signaling.
In Group C, the procedure was completed after access to the retroperitoneal space and exposure of the renal vessels, without performing sympathetic denervation.
In all animals, the colon was subsequently opened, and fecal material was released into the peritoneal cavity in order to induce fecal peritonitis and septic shock according to the criteria. Finally, the abdominal wall was closed with continuous sutures (PDS Plus suture 1, Ethicon J&J Med Tech, Edinburg, Scotland), and the skin was closed in anatomical layers (Prolene suture 2/0, Ethicon J&J Med Tech, Edinburg, Scotland)
During the first phase of the experimental protocol (surgical intervention), animals received intravenous paracetamol (20 mg/kg, Fresenius Kabi Hellas Inc, Athens, Greece) and intramuscular fentanyl (0.05 mg/kg, Kalcex Inc, Riga, Latvia) every 2 hours. During the second phase (septic shock), additional analgesia was administered, including intramuscular morphine (3 mg/kg, Molteni Pharmaceutics Inc, Florence,Italy) and intravenous tramadol (2 mg/kg, Molteni Pharmaceutics Inc, Florence,Italy) as this condition was considered painful and of extremely high severity (15, 16, 17, 18, 19, 20).
Autopsy and Histopathological Analysis
Scheduled euthanasia was performed 6 hours after the induction of fecal peritonitis and septic shock, in accordance with the fundamental principles of laboratory animal euthanasia. Specifically, while remaining under general anesthesia, the pigs received an intravenous overdose of propofol (CP-Pharma Handlesgesellschaft mbH, Burgdorf, Germany).
The previous abdominal incision was reopened, and the thoracic wall was also opened for collection of tissue samples from the liver, lungs, and heart. The kidneys were excised en bloc together with the renal vessels, and tissue samples from the lungs, liver, and heart were collected and fixed in 10% neutral buffered formalin for histological examination. Histological sections from each organ were prepared using standard paraffin embedding techniques. Tissue sections of 4 μm thickness were stained with hematoxylin and eosin (H&E) and evaluated microscopically.
Each tissue section was examined under light microscopy for the presence or absence of pathological lesions. Kidney samples were assessed for acute tubular injury, glomerular damage, interstitial capillary congestion and microhemorrhages. Lung samples were evaluated for alveolar injury, edema, leukocytic infiltration, capillary congestion, atelectasis, and microhemorrhages. Heart samples were examined for edema by measuring the interfibrilar distance, congestion- increase in capillaries, microscopic bleeding and leukocytic infiltration. Liver samples were assessed for capillary congestion, degenerative hepatocellular changes, periportal infiltration and Kupffer cell hyperplasia.
Hemodynamics, monitoring and urine output
During both the surgical procedure and the septic shock phase, hemodynamic parameters were continuously monitored. A sphygmomanometer was used to measure systolic (SBP) and diastolic (DBP) arterial blood pressure, as well as heart rate (HR), with the cuff applied to the thigh of each animal. Measurements were recorded prior to exploratory laparotomy in both groups. In Group R, values were additionally recorded after renal vessel denervation, whereas in Group C measurements were taken after access to the retroperitoneal space. Finally, measurements were obtained after induction of fecal peritonitis and septic shock in both groups. At the same time points, urine output (URINE) was assessed via Foley urinary catheters.
Laboratory Parameters
Each pig underwent a total of three blood samplings: the first prior to laparotomy, the second following completion of the surgical procedure, and the third 6 hours after induction of fecal peritonitis and septic shock just before euthanasia. Approximately 7 mL of blood was collected from a central vein (femoral or jugular) using a 10 mL syringe and transferred into blood collection tubes, which were subsequently sent for laboratory analysis. The measured parameters included White Blood Cell count (WBC) and Platelet count (PLT), coagulation parameters, specifically Prothrombin time (PT), activated Partial Thromboplastin time (aPTT), and International Normalized Ratio (INR). Also biochemical analysis was performed including measurement of Blood Urea Nitrogen (BUN), Creatinine (Creat), Potassium (K), Sodium (Na), hepatic enzymes Aspartate Aminotransferase (AST/SGOT) and Alanine Aminotransferase (ALT/SGPT), and C-reactive protein (CRP).
Statistical Analysis
Sample size calculation was performed using G*Power software version 3.1.9.7, based on data from the international literature. Data are presented as means ± SD. Statistical analysis was performed using R: A Language and Environment for Statistical Computing, version 1.8.4 (R Foundation for Statistical Computing, Vienna, Austria). Normality of data distribution was assessed using the Shapiro–Wilk test. To assess changes over time, linear mixed-effects models were applied with group, time, and their interaction as fixed effects, and subject-specific random intercepts to account for repeated measurements. Within-group changes were studied using estimated marginal means from the mixed-effects models. For qualitative variables, odds ratios (ORs) and expected frequencies (EFs) were calculated. A p value less than 0.05 was considered to be statistically significant.
Results
Table 1 summarizes the baseline demographic, clinical, and laboratory characteristics of the study population. A total of 14 pigs were included, with 9 in the R group and 5 in the C group. Overall, baseline demographic, clinical, and laboratory characteristics were comparable between the R and C groups, with no statistically significant differences observed across all variables (p > 0.05).
Table 1. Baseline demographic, clinical and laboratory characteristics in total population and groups of study.
|
Characteristics |
All (N=14) |
R group (N=9) |
C group (N=5) |
p value |
|
Sex (Male/Female) |
10/4 |
6/3 |
4/1 |
0.266 |
|
Age (months) |
13.7±1.7 |
13.6±1.7 |
14±2 |
0.671 |
|
Weight (kg) |
26.5±1.9 |
26.1±1.9 |
27.2±1.9 |
0.326 |
|
SBP (mmHg) |
118.9±8.8 |
120.2±8.2 |
116.4±10.3 |
0.343 |
|
DBP (mmHg) |
65.2±6.4 |
63±6.3 |
69±4.9 |
0.1 |
|
HR (Beats/min) |
90.9±8.5 |
93±7.7 |
87.4±9.7 |
0.398 |
|
URINE (ml) |
84.6 |
79.4±8.5 |
94±19.2 |
0.097 |
|
WBC (N/μL) |
14241±2656 |
14511±3142 |
13680±1620 |
0.637 |
|
CRP (mg/dL) |
0.078±0.07 |
0.087±0.085 |
0.06±0.025 |
0.561 |
|
PT (sec) |
9.31±2.33 |
9.03±2.31 |
9.82±2.53 |
0.886 |
|
INR |
0.83±0.2 |
0.8±0.2 |
0.88±0.23 |
0.884 |
|
aPTT (sec) |
21±24.9 |
20.2±26.8 |
22.3±24 |
0.921 |
|
PLT (N/μL) |
554571±245886 |
547000±210829 |
568200±327467 |
0.86 |
|
BUN (mg/dL) |
17±5.7 |
15.2±4.5 |
20.2±6.8 |
0.271 |
|
CREA (mg/dL) |
1.05±0.25 |
1.07±0.27 |
1±0.22 |
0.762 |
|
K (mEq/L) |
4±0.62 |
3.8±0.7 |
4.2±0.36 |
0.278 |
|
Na (mEq/L) |
135±6.7 |
136±6.8 |
132±6.2 |
0.711 |
|
SGOT (U/L) |
58.6±30.7 |
56.5±31.5 |
62.2±32.5 |
0.834 |
|
SGPT (U/L) |
63.2±19.9 |
60±19 |
68.4±22.6 |
0.772 |
SBP: systolic blood pressure, DBP: diastolic blood pressure, HR: heart rate, URINE: urine output, WBC: white blood cell, CRP: C-reactive protein, PT: prothrombin time, INR: international normalized ratio, aPTT: partial thromboplastine time, PLT: platelet count, BUN: blood urea nitrogen, CREA: creatinine, K: potassium, Na: sodium, SGOT: aspartate aminotransferase (AST), SGPT: alanine aminotransferase (ALT). Values are mean ± SD. R group: renal denervation, C group: control.
Systemic hemodynamic measurements
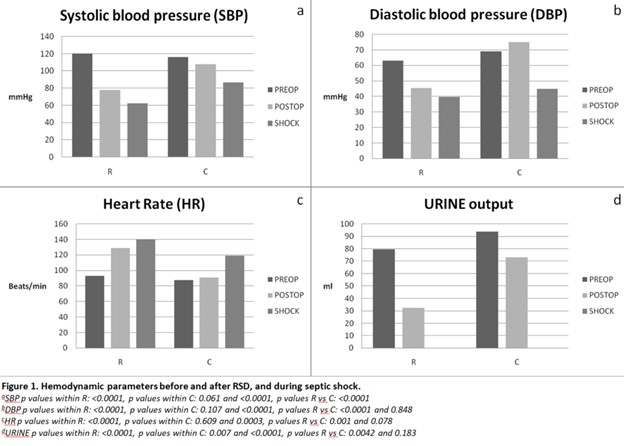
Renal sympathetic denervation causes a strong, immediate, and sustained drop in both systolic (SBP) and diastolic (DBP) blood pressure in pigs of R group. Systolic blood pressure (SBP) decreased significantly immediately after surgery in denervated pigs (p < 0.001) and continued to drop at 6 hours post-operatively (p < 0.001) Compared to control group, the post-operative SBP decrease in R pigs was significant (p < 0.001). Similarly, DBP decreased significantly immediately after surgery in denervation group (p < 0.001) and further at shock time (p < 0.001). Compared to control group, DBP in R group was significantly lower immediately postoperatively (p < 0.001). However, after the induction of shock the two groups had no statistically significant difference (p = 0.848).
Renal denervation strongly affects heart rate (HR), with an immediate and sustained increase, both within the R group and relative to control animals at 6 hours postoperatively. Heart Rate (HR) increased significantly immediately after surgery in denervated pigs (p < 0.0001) and continued to rise 6 hours postoperatively (p < 0.001). Nevertheless, at shock time point the increase in R pigs was not significantly different compared to control group, (p = 0.078).
Moreover, denervation induces a dramatic and sustained decrease in urine output in R pigs, most pronounced at shock time point. Urine output in R group decreased significantly immediately after surgery (p < 0.001) and further at 6 hours post-operatively (p < 0.001) Compared to control animals, urine output was significantly lower in R group immediately after operation (p <0.0042) but as opposed to 6 hours time point when two groups did not show statistically significant difference (p = 0.183) (Figure 1).
Inflammatory biomarkers
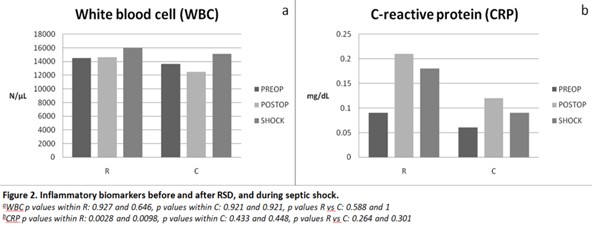
White blood cell count (WBC) did not change significantly after surgery in the denervated pigs (p> 0.5) and there were no significant differences compared to control group at any time point (p > 0.5). On the other hand, renal denervation caused a clear and statistically significant increase in CRP in R group, both immediately (p = 0.0028) and 6 hours after the shock induction (p = 0.0098). Although CRP increased in the R group, the change was not significantly larger than the control group at any time point (p > 0.26) (Figure 2).
Coagulation system markers
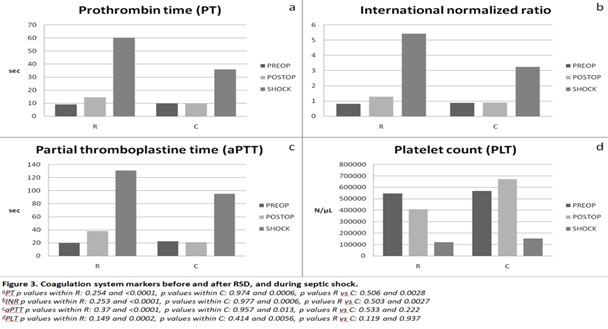
Renal denervation caused a delayed and strong increase in PT in the R group, but no immediate change was observed. Prothrombin Time (PT) did not change immediately after surgery in the operated pigs (p = 0.254), but increased markedly 6 hours post-operatively (p < 0.001). The denervation effect is evident compared to control group after shock-induction, meaning this increase is likely attributable to the renal sympathetic denervation rather than baseline fluctuations. The 6h postoperatively increase was significantly larger than in control animals (p = 0.0028), indicating a denervation-specific effect on PT.
Equivalent was the observed effect on INR, which did not change immediately after surgery in denervated animals (p = 0.253), but increased significantly 6 hours postoperatively (p < 0.001). The increase at shock time was significantly larger than in the control group (p = 0.0027), indicating an effect on coagulation system. As for the impact on Partial Thromboplastin Time (aPTT), this marker did not change immediately after surgery in R group (p = 0.37), but increased significantly at shock time point (p < 0.001). However, this time point increase was not significantly different from control group (p = 0.22), though the magnitude suggests a biologically meaningful effect. Lastly, Platelets count (PLT) did not change immediately after surgery in R group (p = 0.15), but decreased significantly 6 hours postoperatively (p = 0.0002), indicating a delayed thrombocytopenic response. However, the drop was not statistically different from control group either immediately postoperatively or at shock time (p > 0.11), likely due to small sample size or variability in controls, although the magnitude suggests a clear denervation-associated decrease (Figure 3).
Renal function and Electrolytes biomarkers
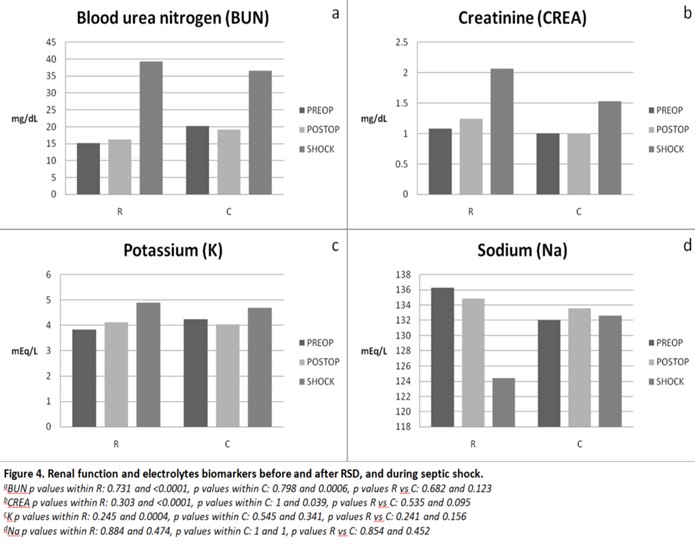
The effect of renal denervation on renal function in septic shock was based on Blood Urea Nitrogen (BUN) and Creatinine (CREA) measurements. BUN did not change immediately after surgery in the group of denervated pigs (p = 0.731), but increased significantly after the shock induction (p < 0.001). However, this reported increase was not significantly different from the control group (p > 0.12). The renal denervation effect on creatinine (CREA) appears delayed, showing up only at 6 hours postoperatively in the R group (p < 0.001). Although creatinine increased in the R group at the 6 hours after surgery time point, the change was not significantly larger than any change seen in control animals (p = 0.095).
Moreover, potassium (K) and sodium (Na) measurements took place in both groups in order to study the effect on electrolytes in septic shock. Potassium (K) did not change immediately after surgery in denervated animals (p = 0.245), but increased significantly at 6 hours time point of shock (p = 0.0004). However, the increase was not significantly different from the control group (p > 0.15). Accordingly, Sodium (Na) did not change immediately or 6 hours after surgery at shock time in the R group of renal denervation (p= 0.884 and p= 0.474, respectively). There were no significant differences compared to the control group at any time point (p > 0.45) (Figure 4).
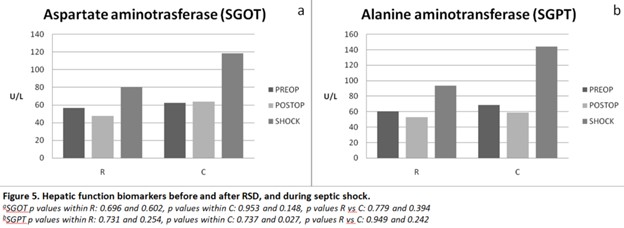
Hepatic function biomarkers
Hepatic function was based on SGOT and SGPT measurements as depicted on Figure 5, respectively. Neither SGOT nor SGPT were affected by renal denervation in this dataset. SGOT (AST) did not change immediately or 6 hours after surgery in R group (p> 0.6). There were no significant differences compared to control group at any time point (p > 0.39). Likewise, SGPT (ALT) did not change immediately after denervation or at shock time in the R group (p= 0.731 and p= 0.254, respectively). Similarly, there were no significant differences compared to the control group at any time point (p > 0.24).
Analysis of alterations over time
Additionally, renal denervation effects on laboratory and clinical parameters were evaluated by comparing the change of values from preoperative to induction of shock between the R (denervation) and the C (control) groups (Table 2).
Table 2: Changes from baseline to septic shock.
|
|
Group |
Alteration to shock |
p value to shock |
p value R vs C |
|
|
SBP (mmHg) |
R |
-57.67 |
<0.001 |
|
|
|
C |
-29.6 |
0.00043 |
0.003 |
||
|
|
|
|
|
|
|
|
DBP (mmHg) |
R |
-23.33 |
<0.001 |
|
|
|
C |
-30.2 |
0.001 |
0.78 |
||
|
|
|
|
|
|
|
|
ΗR (Beats/min) |
R |
+47.56 |
<0.001 |
|
|
|
C |
+31.6 |
0.0008 |
0.099 |
||
|
|
|
|
|
|
|
|
URINE (ml) |
R |
-70.56 |
<0.001 |
|
|
|
C |
-94 |
0.0003 |
0.049 |
||
|
|
|
|
|
|
|
|
WBC (N/μL) |
R |
+1463 |
0.398 |
|
|
|
C |
+1460 |
0.453 |
0.78 |
|
|
|
|
|
|
|
|
|
|
CRP (mg/dL) |
R |
+0.092 |
0.0185 |
|
|
|
C |
+0.034 |
0.246 |
0.233 |
|
|
|
|
|
|
|
|
|
|
PT (sec) |
R |
+51.13 |
<0.001 |
|
|
|
|
C |
+26.24 |
0.003 |
0.016 |
|
|
|
|
|
|
|
|
|
INR |
R |
+4.61 |
<0.001 |
|
|
|
|
C |
+2.37 |
0.211 |
0.015 |
|
|
|
|
|
|
|
|
|
aPTT (sec) |
R |
+110.68 |
<0.001 |
|
|
|
|
C |
+72.86 |
0.016 |
0.386 |
|
|
|
|
|
|
|
|
|
PLT (N/μL) |
R |
-427444 |
0.00026 |
|
|
|
|
C |
-415200 |
0.037 |
0.78 |
|
|
|
|
|
|
|
|
|
BUN (mg/dL) |
R |
+24.11 |
<0.001 |
|
|
|
|
C |
+16.4 |
0.018 |
0.38 |
|
|
|
|
|
|
|
|
|
CREA (mg/dL) |
R |
+0.993 |
<0.001 |
|
|
|
|
C |
+0.532 |
0.18 |
0.41 |
|
|
|
|
|
|
|
|
|
K (mEq/L) |
R |
+1.06 |
0.00047 |
|
|
|
|
C |
+0.46 |
0.211 |
0.58 |
|
|
|
|
|
|
|
|
|
Na (mEq/L) |
R |
-11.89 |
0.355 |
|
|
|
|
C |
+0.6 |
0.927 |
0.8 |
|
|
|
|
|
|
|
|
|
SGOT (U/L) |
R |
+23.78 |
0.389 |
|
|
|
|
C |
+56.40 |
0.246 |
0.581 |
|
|
|
|
|
|
|
|
|
SGPT (U/L) |
R |
+33.33 |
0.226 |
|
|
|
|
C |
+75.60 |
0.18 |
0.58 |
|
SBP: systolic blood pressure, DBP: diastolic blood pressure, HR: heart rate, URINE: urine output, WBC: white blood cell, CRP: C-reactive protein, PT: prothrombin time, INR: international normalized ratio, aPTT: partial thromboplastine time, PLT: platelet count, BUN: blood urea nitrogen, CREA: creatinine, K: potassium, Na: sodium, SGOT: aspartate aminotransferase (AST), SGPT: alanine aminotransferase (ALT). Values are mean ± SD. R group: renal denervation, C group: control.
Significant differences between groups were observed for several variables:
For other parameters, including WBC, PLT, CRP, BUN, CREA, PTT, HR, SGOT, SGPT, K, and Na, no statistically significant difference was observed between groups over the 6-hour post-operative period (all p > 0.05).
Microscopic findings and histopathological analysis
Table 3 represents the pathological lesions found microscopically in tissue sections after histopathological analysis. No lesion differed significantly between groups after correction for multiple statistical testing. However, directional trends were observed, with pulmonary injury in R group of denervation and reduced hepatic degenerative changes compared to controls. In kidney, glomerular damage was more frequent in C group (80%) rather than R group (33%), but no statistically significantly difference observed (p= 0.265). In liver, R group had much lower degenerative changes (22%) compared to control group (80%), although the difference was not statistically significant (p= 0.09). Histopathology of lung tissue revealed higher atelectasis in R group (89%) compared to C group (40%) and, also, higher leukocytic infiltration in R group (78%) rather than C group (20%), but none was statistically significantly different (p= 0.094 and p= 0.09, respectively). Additionally, denervated animals manifested more frequent alveolar damage but not statistically different than C group (p= 0.265). Myocardium examination had no meaningful group separation.
Table 3: Histopathological analysis.
|
Tissue |
Pathological lesion |
Groups |
P value |
|
|
R |
C |
|||
|
Kidney |
Acute Tubular Injury |
78% |
80% |
1 |
|
Glomerular Damage |
33% |
80% |
0.265 |
|
|
Interstitial Capillary Congestion |
100% |
100% |
1 |
|
|
Microhemorrages |
78% |
60% |
0.58 |
|
|
Lung |
Alveolar Damage |
78% |
40% |
0.265 |
|
Edema |
33% |
40% |
1 |
|
|
Leukocytic Infiltration |
78% |
20% |
0.09 |
|
|
Capillary Congestion |
100% |
100% |
1 |
|
|
Atelectasis |
89% |
40% |
0.094 |
|
|
Microhemorrages |
56% |
20% |
0.3 |
|
|
Heart |
Edema- Interfibrilar distance |
78% |
80% |
1 |
|
Capillary Congestion |
100% |
80% |
0.357 |
|
|
Microscopic Bleeding |
44% |
60% |
1 |
|
|
Leukocytic Infiltration |
33% |
20% |
1 |
|
|
Liver |
Capillary Congestion |
78% |
100% |
0.5 |
|
Degenerative Changes |
22% |
80% |
0.09 |
|
|
Portal Tract Inflammation |
56% |
80% |
0.58 |
|
|
Kupffer Cell Hyperplasia |
67% |
100% |
0.258 |
|
Discussion
The main findings of the study were that surgical bilateral renal sympathetic denervation affected hemodynamic response and coagulation system after sepsis- induce shock by fecal peritonitis in pigs. Renal failure obtained by septic shock was not influenced by bilateral denervation. On the other hand, inflammatory response and hepatic function remained totally unaffected.
Previous studies of experimental porcine models pointed out the hemodynamic instability as well as the renal and hepatic failure after occurrence of septic shock. (21, 22) Hemodynamic changes are presented by increase of heart rate and drop in blood pressure, systolic and diastolic. Renal failure is confirmed by increased values of plasma creatinine and hyperkalaemia accompanied by decrease of urine output.(11, 23, 24) The negative impact on hepatic function was validated by the increase of serum concentrations of hepatic enzymes such as alanine aminotransferase (ALT) and aspartate aminotransferase (AST) (22). Notably, porcine experimental models are very close to humas because of their kidney similarity and organic response rather than rats, mice or dogs (23). Thus, selection of porcine models could lead to exporting safer conclusions regarded to human organism. Moreover, according to previous studies, the organ dysfunction due to septic shock is depicted and confirmed by several histopathological changes after the microscopic evaluation of the tissue sections and histopathological analysis. The most serious changes in kidney are acute tubular injury, glomelural damage, interstitial capillary congestion as result of hemodynamic and inflammatory factors combined with sepsis. Lung parenchyma because of sepsis develops mostly edema, atelectasis, alveolar damages, capillary congestion and inflammation such as leucocytic infiltration. Myocardium is also affected microscopically during shock condition by developing capillary congestion, leucocytic infiltration and edema depticted by the interfibrilar distance. Septic liver histopathology is characterized by portal tract inflammation, kupffer cell hyperplasia, degenerative changes and capillary congestion. Lastly, the influence of sepsis on coagulation system is confirmed by the microscopic bleeding and microhemorrages found after histopathologic evaluation of all organs involved (11, 25, 26).
In our study preoperative and 6h after induction of septic shock values in each group, denervated (R) and control (C) and for all measured parameters were compared in order to evaluate the progression and impact of septic shock in organic response. Actually, renal failure was confirmed by the statistically significant increase of Blood Urine Nitrogen (BUN) and creatinine (CREA) as well as the decrease of urine output (URINE) in both groups. Hemodynamic change and instability of septic shock was also obtained because of the significant drop of Systolic (SBP) and Diastolic (DBP) blood pressure accompanied by highly significant increase of Heart Rate (HR). However, there were no significant changes in values of hepatic enzymes, SGOT/ AST and SGPT/ALT, in both groups 6h after the shock occurrence resulting in no impact on hepatic function as previous studies. Moreover, statistically significant lhyperkalaemia occurred in denervated animals but sodium was not affected by the progression of sepsis in both groups.
The impact on coagulation system consist of a territory never evaluated before in porcine septic model, using the parameters of Prothrombin time (PT), International Normalized Ratio (INR), Partial thromboplastin time (aPTT) and Platelet count (PLT). In total porcine population a significant increase of clotting factors in addition to decrease of platelet count was observed as the time passed and septic shock installed. These findings keep up with the expected coagulopathy in sepsis due to microvascular endothelial injury leading to fibrinolysis and coagulation imbalance (27).
Lastly, C-reactive protein (CRP) and White Blood Cell count (WBC) were measured in order to study the effect on inflammatory response. Occurrence of sepsis affected CRP in the denervation group by increasing it significantly, without, however, leading to the same result as for the WBC.
According to previous studies referring to renal sympathetic denervation in swine models and the systemic impact of the procedure, several changes have been reported. Dong Won Lee et al concluded that RSD did not affect renal function parameters such as plasma blood urea nitrogen (BUN) and creatinine (CREA), electrolytes such as potassium (K) and Sodium (Na) and inflammatory marker C-reactive protein (CRP). However, proinflammatory (IL-1b, IL-18), inflammatory (IL-6, TNF-a) and anti-inflammatory (IL-10) cytokines increased immediately after RSD (28).
Other studies revealed significant outcomes in hemodynamics immediately after RSD by decrease in SBP, DBP and mean arterial pressure (MAP) in pigs (29, 30). Moreover, Dan Li et al, reported significant histological effect of RSD presented as vascular wall and/or renal parenchyma damage with greater number of lesions, thrombotic masses and kidney parenchyma hemorrhage, in 92% of studied animals (29). Verloop et al, observed histological changes with severe inflammation response, neural degeneration and reduction in markers expression in a porcine model of renal denervation (31).
Other porcine model studies concluded on acute improvement of renal hemodynamic directly after RSD by increasing renal blood flow (RBF), glomerular filtration rate (GFR) and average peak velocity (APV) (14, 31, 32,).
On the other hand, in rat models of renal denervation, RSD improved arterial hypertension, cardiac function and cortical blood flow (CBF). However, medullar blood flow (MBF) and GFR remained unaffected. Moreover, arterial fibrosis decreased but not atrophy or apoptosis in kidney (33, 34).
In our study, took place a comparison between preoperative and immediately after operation values for each of 16 parameters for both groups of animals. Renal sympathetic denervation affected the hemodynamic profile on R group reporting a statistically significant drop of SBP, DBP, HR and urine output. Also a significant increase of inflammation was observed in the same group because of the increase of CRP. All the other parameters such as clotting agents (PT, aPTT, INR, PLT), renal function markers (BUN, CREA), hepatic markers (SGOT, SGPT), electrolytes (K, Na) and WBC did not show a remarkable change in the group of denervation. On the other hand, in the control group none of the parameters obtained a significant change immediately after operation.
Comparison of group R and control animals directly after operation revealed a significant effect of renal denervation only on hemodynamic condition reporting significant drop of blood pressure either systolic or diastolic and significantly higher heart rate.
The impact of bilateral sympathetic denervation of renal nerves in systemic response in septic shock condition constitutes a subject that very few experimental studies dealt with. Among these studies none used swine as experimental model, an animal more similar to human organism by the prism of anatomy and architecture, urinary concentration, tolerance in tissue hypoxia and ischemia occurrence (23).
In a sheep model of experimental hyperdynamic sepsis, renal denervation caused significant higher decrease in blood pressure, drop in urine output and increase in plasma craetinine compared to control group. However, both groups had recorded similar increase in heart rate and renal blood flow. The study concluded that renal sympathetic nerves and their activity alterations do not influence the development of acute kidney injury during septic shock (1).
Similar experimental protocol, resulted in higher renal blood flow and cortical perfusion without effect on kidney medullar perfusion after renal denervation. Moreover, the study reported no significant difference in increase of creatinine or decrease of urine output and drop of mean arterial pressure (MAP) between denervated and control animals. In summary, renal function and hypoperfusion, metabolic changes during septic shock and survival remained unaffected by renal denervation (12).
Wang et al, revealed a protective effect of renal denervation in mice after endotoxemia-induced acute renal failure because of the decrement in GFR and increase of RBF obtained in mice with denervated kidneys (13).
In our study, renal denervation led to a marked and sustained decrease of systolic blood pressure during septic shock. On the other hand, all the other hemodynamic measurements (diastolic blood pressure, heart rate and decrement of urine output) were not statistically different compared to control group. These results reveal a low hemodynamic impact of renal denervation in sepsis-induced shock.
Moreover, surgical denervation seemed to affect the coagulation system after induction of sepsis because of the significant increase in clotting markers such as PT and INR compared to control group at the same time point. On the other hand, although denervated pigs showed a significant rise of aPTT and sharp drop in platelet count, these findings did not differ statistically from control fluctuations.
Referring to renal function, denervation induces a measurable delayed increase in BUN and CREA in R group but these changes are not definitively different from control group. Concluding that progression of renal failure in shock does not depend on sympathetic nerve activity.
As about the inflammatory markers, surgery did not have a detectable effect on WBC. On the other hand, it caused a clear and statistically significant increase of CRP in R group but this change was not significantly larger than control group. So that renal denervation did not affect the inflammatory response in septic shock.
Although potassium rises significantly at shock time point in R pigs, the difference compared to control group is not significant. As for the remaining parameters, sodium, SGOT and SGPT denervation did not to have any effect as the changes in R group did not have significant differences from control group after induction of shock. These results indicate that surgery did not affected metabolic or hepatic activity and progression in septic shock condition.
Moreover, according to previous studies, the organ dysfunction due to septic shock is depicted and confirmed by several histopathological changes after the microscopic evaluation of the tissue sections and histopathological analysis. The most serious changes in kidney are acute tubular injury, glomelural damage, interstitial capillary congestion as result of hemodynamic and inflammatory factors combined with sepsis. Lung parenchyma because of sepsis develops mostly edema, atelectasis, alveolar damages, capillary congestion and inflammation such as leucocytic infiltration. Myocardium is also affected microscopically during shock condition by developing capillary congestion, leucocytic infiltration and edema depticted by the interfibrilar distance. Septic liver histopathology is characterized by portal tract inflammation, kupffer cell hyperplasia, degenerative changes and capillary congestion. Lastly, the influence of sepsis on coagulation system is confirmed by the microscopic bleeding and microhemorrages found after histopathologic evaluation of all organs involved (11, 25, 26).
In present study, histopathological analysis revealed widespread organ injury consistent with septic shock. Although examination of pathological lesions had no statistical significant difference between groups, directional trends were observed in liver, kidney and lung. The strongest directional signal trend was towards less parenchymal liver damage in renal denervation group. Moreover, renal denervation relates to worse pulmonary injury because of the higher atelectasis and leukocytis infiltration meaning worsen lung collapse and inflammatory injury. Lastly, renal denervation led to less glomerular damage in kidneys revealing a possible protective trend after septic shock occurrence. In myocardium pathological lesions developed similarly so remained unaffected by renal sympathetic denervation.
Conclusion
In this porcine model of fecal peritonitis–induced hyperdynamic septic shock, bilateral renal sympathetic denervation produced immediate and sustained alterations in cardiovascular homeostasis and coagulation function, evidenced by significant perioperative hypotension and marked prolongation of PT and INR during shock. Despite these hemodynamic and hematologic effects, renal denervation did not mitigate the progression of septic acute kidney injury nor influence the systemic inflammatory, hepatic, or electrolyte disturbances associated with septic shock. Histopathological evaluation similarly revealed no significant intergroup differences in organ injury severity.
Collectively, these findings indicate that renal sympathetic signaling contributes to circulatory and coagulation responses during septic shock, yet its interruption is not associated with increased risk in sepsis. Further research with larger cohorts and extended observation periods is warranted to clarify the mechanistic role of renal sympathetic pathways in sepsis and to determine whether timing or adjunctive interventions could modify these outcomes.
Strengths and Limitations
Strengths of the present study were the successfully performed bilateral renal sympathetic denervation as well as the selection of porcine experimental model which could lead to conclusions more related to human organism. However, several limitations should be mentioned. The limited number of animals could possibly be related to underpowered conclusions of the study, even though it could become the first step for further investigation. Moreover, traditional parameters, such as blood urea nitrogen, creatinine and liver enzymes, have several limitations to predict an early stage of renal and hepatic failure, respectively.
Acknowledgments
The authors acknowledge Bratko Ilias, Vasiliki Tsakiri, Savvaidis Panagiotis, Skriapa Athina and Mitropoulos Athanasios for their contribution to this work.