International Journal of Epidemiology And Public Health Research
OPEN ACCESS | Volume 9 - Issue 1 - 2026
ISSN No: 2836-2810 | Journal DOI: 10.61148/2836-2810/IJEPHR
Magoba Ronald Arnold1*, Mogere Dominic2, Magu Dennis3, Mudenyo Mark Owang4
1Tuberculosis Management Center, Kenyatta National Hospital (KNH), Nairobi, Kenya. 2Mount Kenya University –School of Public Health, Thika, Kenya.
3Field Epidemiology and Laboratory Training Program, Ministry of Health, Nairobi, Kenya.
4Respiratory and Infectious Disease Unit, Kenyatta National Hospital, Nairobi, Kenya.
*Corresponding author: Magoba Ronald Arnold, Tuberculosis Management Center, Kenyatta National Hospital (KNH), Nairobi, Kenya.
Received: March 10, 2026 | Accepted: March 22, 2026 | Published: April 08, 2026
Citation: Magoba R Arnold, Dominic M, Dennis M, Mudenyo M Owang., (2026) “Determinants And Consequences of Patient and Health System Delays in Tuberculosis Diagnostics and Treatment Among Individuals Aged 15 Years and Above at Kenyatta National Hospital, Nairobi County, Kenya”. International Journal of Epidemiology and Public Health Research, 9(2); DOI: 10.61148/2836-2810/IJEPHR/192.
Copyright: © 2026. Magoba Ronald Arnold. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Background: Delays in tuberculosis (TB) diagnosis and treatment continue to compromise control efforts, particularly in high-burden settings such as Kenya. These delays increase transmission, worsen clinical outcomes, and elevate mortality. The post-COVID-19 period has further strained health- care systems, influencing patient health-seeking behaviour and diagnostic efficiency. Objective: To quantify patient-related and health-system-related delays in TB diagnosis and treatment and to identify key determinants of these delays among adults receiving care at Kenyatta National Hospital (KNH). Methods: A descriptive cross-sectional study involving 128 smear- positive pulmonary TB patients was conducted in 2022. Participants aged
≥15 years were selected through systematic sampling. Data on socio- demographic factors, care-seeking pathways, and diagnostic intervals were collected through structured interviews. Delay durations were categorized using the median as the cut-off point. Descriptive statistics, chi-square tests, and logistic regression were used to determine predictors of patient and system delays. Statistical significance was set at p < 0.05. Results: Women constituted 51.2% of participants, with the largest age group being 40–49 years (32.3%). A substantial proportion (29.9%) sought medical care only after 2–3 months of symptoms. Distance to health facilities, socio-economic constraints, and health-worker-related factors were significantly associated with prolonged delay (p < 0.05). Many patients (66.9%) reported fear upon receiving a diagnosis, while 96.1% believed they should access treatment free of charge. Perceived community stigma was high (96.9%), and both patient behaviour and health-system processes were considered contributors to delayed care. Conclusion: Significant patient-level and system-level delays persist at KNH, highlighting critical gaps in early TB detection and timely initiation of treatment. Strengthening diagnostic capacity, decentralizing services, enhancing patient awareness, and improving health- worker responsiveness are essential to accelerate TB case detection and align with national and global TB-control targets.
Tuberculosis, Diagnostic delay, Patient delay, Health-system delay, Kenya, Epidemiology
1. Introduction
Tuberculosis (TB) remains one of the world’s most persistent public-health challenges and continues to impose a disproportionate burden on low- and middle-income countries. Despite decades of progress in diagnostics, treatment, and prevention, the World Health Organization (WHO) reported an estimated 10.3 million new TB cases and 1.3 million associated deaths in 2023. More than 85% of these cases occurred in resource-limited settings, where constrained health-system capacity and socioeconomic vulnerabilities hinder timely detection and effective treatment. Kenya remains one of the 30 globally designated high-TB-burden countries, with an estimated incidence of 256 cases per 100,000 people in 2023.
Timely diagnosis is fundamental to interrupting TB transmission, improving clinical outcomes, and preventing progression to severe disease. However, delays in diagnosis and treatment initiation continue to threaten the effectiveness of TB-control programs worldwide. Patient-related delays—often caused by limited awareness of symptoms, financial constraints, stigma, or reliance on self-medication—allow continued transmission within households and communities. Health-system delays, which include prolonged diagnostic pathways, inadequate laboratory capacity, and inefficiencies within referral systems, further compound the problem. Together, these delays contribute substantially to TB-related morbidity, mortality, and the emergence of drug-resistant strains.
The COVID-19 pandemic further disrupted TB detection and treatment services, reversing several years of progress. Lockdowns, overwhelmed health facilities, transport disruptions, and fear of contracting COVID-19 discouraged care-seeking and reduced diagnostic throughput globally. In 2020 alone, TB case detection dropped by nearly 25% worldwide. Although Kenya has made strides in restoring essential services, the residual effects of the pandemic are still being felt, particularly within tertiary hospitals that manage large and complex patient populations.
Kenyatta National Hospital (KNH), Kenya’s largest referral and teaching hospital, receives substantial numbers of patients with advanced or complicated TB. The hospital’s high patient load, coupled with intricate referral pathways, can extend the time between symptom onset, diagnosis, and initiation of treatment. Understanding the magnitude of delays and the factors driving them in such a large tertiary hospital is critical, as delayed care at this level may influence outcomes for patients referred from across the country.
Previous studies in Kenya have documented delays in TB diagnosis in primary and secondary health facilities; however, limited evidence exists from tertiary centres such as KNH, especially during the post-COVID-19 recovery period. Given the unique patient demographics, referral patterns, and service delivery pressures at KNH, a context-specific analysis is needed to identify key determinants of delay and guide targeted interventions.
This study therefore assessed the duration and determinants of patient-related and health-system-related delays among adults diagnosed with pulmonary TB at KNH. The findings aim to support improved case-detection strategies, strengthen diagnostic and referral systems, and inform national TB-control priorities in line with the WHO End TB Strategy.
2. Methods and Materials
2.1 Study Design and Setting
This study employed a descriptive cross-sectional design to assess patient-related and health-system-related delays in tuberculosis (TB) diagnosis and treatment. The study was conducted at Kenyatta National Hospital (KNH), located in Nairobi County, Kenya. KNH is the country’s largest national referral, teaching, and research hospital, with a bed capacity exceeding 2,000 and serving more than 10,000 clients daily. The hospital diagnoses an estimated 200–300 TB cases every month and provides diagnostic and treatment services in line with national and WHO-recommended TB management guidelines. All testing and treatment services are offered free of charge.
2.2 Study Population
The target population comprised adults aged 15 years and above who were newly diagnosed with smear-positive pulmonary TB at KNH. Eligible participants were those who had been on anti-TB treatment for less than six months at the time of recruitment. Individuals with a history of previous TB treatment (retreatment cases), patients diagnosed with extrapulmonary TB, and those residing outside Nairobi County were excluded from the study.
2.3 Sampling Procedure and Sample Size
Systematic sampling was used to recruit participants from patient registration records at the KNH TB clinic. The estimated number of newly diagnosed TB patients presenting during the study period was divided by the required sample size (n = 128) to determine the sampling interval (K). A random number between 1 and K was selected as the starting point, and every Kth eligible patient was subsequently enrolled until the required sample size was achieved. Once selected, patients were contacted for follow-up interviews to obtain additional information not captured in the clinic records.
2.4 Data Collection Tools and Procedures
Data were collected using a structured, interviewer-administered questionnaire developed specifically for this study. The tool captured:
Recruitment and data collection were done during routine clinic days (Monday to Friday). Each participant was approached after receiving clinical services and invited to participate in the study. Interviews were conducted in a private room to ensure confidentiality. Each session lasted approximately 10–20 minutes. Written informed consent was obtained from all participants.
2.5 Study Variables
2.6 Data Management and Analysis
Completed questionnaires were reviewed for completeness, coded, and double-entered into Epi Info version 7 before being exported to SPSS version 25 for analysis. Descriptive statistics were used to summarize categorical variables using frequencies and percentages, while continuous variables were summarized using medians and interquartile ranges.
The median value of each delay category was used as the cut-off point to classify participants into “delayed” and “not delayed” groups. Associations between categorical variables and delay categories were examined using chi-square (χ²) tests. Variables with a significance level of p < 0.10 in bivariate analysis were retained for multivariable logistic regression. Adjusted odds ratios (AORs) and 95% confidence intervals (CIs) were used to identify independent predictors of delay. A p-value < 0.05 was considered statistically significant. Results were presented using tables, bar charts, and frequency distributions.
2.7 Ethical Considerations
Ethical approval was obtained from relevant institutional review boards. Permission to conduct the study at KNH was granted by the hospital administration. Participation was voluntary, and participants were informed of their right to withdraw at any time without any consequences. All collected data were anonymized and stored securely to maintain confidentiality.
3.0 Results
3.1 Socio-Demographic Characteristics
Although the calculated sample size was 128 participants, one questionnaire was excluded due to incomplete responses. Therefore, a total of 127 participants were included in the final analysis.
A total of 127 adults diagnosed with smear-positive pulmonary tuberculosis participated in the study. Females comprised 51.2% (n = 65), while males accounted for 48.8% (n = 62). The age distribution showed that most patients were between 40–49 years (32.3%, n = 41), followed by 30–39 years (21.3%, n = 27) and 50–59 years (17.3%, n = 22). Only 3.1% (n = 4) were aged 70–79.
Regarding marital status, 43.3% (n = 55) were cohabiting, 29.1% (n = 37) were single, 13.4% (n = 17) were divorced or separated, and 14.2% (n = 18) were widowed. Most respondents had primary or secondary education (59.1%, n = 75), while 26.8% (n = 34) had no formal education and only 14.2% (n = 18) had advanced-level or college education.
Occupation status showed a predominance of independent or self-employed participants (36.2%, n = 46) and professionals (32.3%, n = 41). Unemployment accounted for 13.4% (n = 17). Nearly half the participants (48.8%, n = 62) lived in households with two children.
3.2 TB Care Pathway and Diagnostic Experiences
Government facilities were the primary point of diagnosis, with 92.9% (n = 118) of participants first obtaining their TB diagnosis in public hospitals. Only 7.1% (n = 9) were diagnosed in private facilities. Most respondents (98.4%, n = 125) were already on anti-TB treatment at the time of the survey.
Psychological and social responses were profound: 66.9% (n = 89) felt scared upon diagnosis, while an overwhelming 96.9% (n = 123) perceived TB-related discrimination in their communities. Similarly, 96.1% (n = 122) believed that TB medication should be free.
3.3 Patient Delay: Time from Symptom Onset to First Health-Facility Visit
Patient delay was substantial. The highest proportion of participants (29.9%, n = 38) sought care only after 2–3 months of onset of symptoms, followed by 20.5% (n = 26) who sought care within the first month. Notably, 33.1% (n = 42) of respondents delayed for five months or longer, with 14.2% (n = 18) delaying for more than seven months.
3.4 Associations Between Participant Characteristics and Delays
Chi-square tests identified several statistically significant associations:
These findings suggest that geographic, demographic, and family-context factors strongly influence health-seeking behaviour and TB diagnosis pathways.
Table 1. The socio-demographic characteristics of the participants are summarized in Table 1 (n = 127)
|
Variable |
Category |
n |
% |
|
Sex |
Female |
65 |
51.2 |
|
Male |
62 |
48.8 |
|
|
Age group |
10–19 |
6 |
4.7 |
|
20–29 |
18 |
14.2 |
|
|
30–39 |
27 |
21.3 |
|
|
40–49 |
41 |
32.3 |
|
|
50–59 |
22 |
17.3 |
|
|
60–69 |
9 |
7.1 |
|
|
70–79 |
4 |
3.1 |
|
|
Marital status |
Cohabiting |
55 |
43.3 |
|
Single |
37 |
29.1 |
|
|
Divorced/Separated |
17 |
13.4 |
|
|
Widow |
18 |
14.2 |
|
|
Education |
Advanced/College |
18 |
14.2 |
|
Primary–Secondary |
75 |
59.1 |
|
|
No formal education |
34 |
26.8 |
|
|
Occupation |
Professional |
41 |
32.3 |
|
Independent/Self-employed |
46 |
36.2 |
|
|
Temporary |
11 |
8.7 |
|
|
Housewife |
12 |
9.4 |
|
|
Unemployed |
17 |
13.4 |
|
|
Household size (children) |
0 |
9 |
7.1 |
|
1 |
27 |
21.3 |
|
|
2 |
62 |
48.8 |
|
|
3 |
20 |
15.7 |
|
|
4 |
5 |
3.9 |
|
|
5 |
3 |
2.4 |
|
|
6+ |
1 |
0.8 |
|
|
Socio-Demographic Characteristics A total of 127 adults diagnosed with smear-positive pulmonary tuberculosis participated in the study. Females comprised 51.2% (n = 65), while males accounted for 48.8% (n = 62). The age distribution showed that most patients were between 40–49 years (32.3%, n = 41), followed by 30–39 years (21.3%, n = 27) and 50–59 years (17.3%, n = 22), as illustrated in Figure 1. Only 3.1% (n = 4) were aged 70–79 years. Regarding marital status, 43.3% (n = 55) were cohabiting, 29.1% (n = 37) were single, 13.4% (n = 17) were divorced or separated, and 14.2% (n = 18) were widowed. Most respondents had primary or secondary education (59.1%, n = 75), while 26.8% (n = 34) had no formal education and only 14.2% (n = 18) had advanced-level or college education. Occupational status showed a predominance of independent or self-employed participants (36.2%, n = 46) and professionals (32.3%, n = 41). Nearly half of the participants (48.8%, n = 62) lived in households with two children. Detailed socio-demographic characteristics are presented in Table 1. Sex distribution of participants is shown in Figure 2, with a near-equal representation of males and females.
|
|
|
|
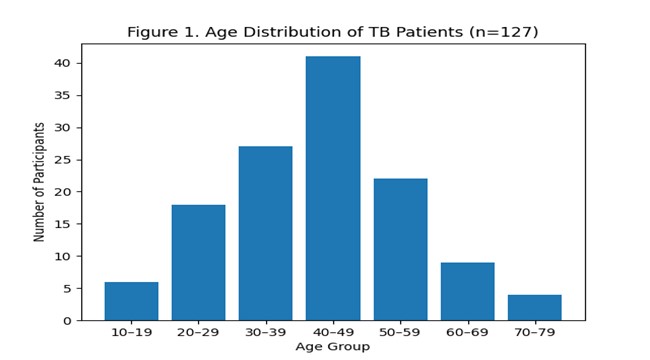
Figure 2 illustrates the distribution of the number of children per household among tuberculosis patients. Most participants reported having between two and four children, reflecting typical household structures within the study population.
Table 2. Care pathway and psychosocial experiences (n = 127)
|
Variable |
Category |
n |
% |
|
Initial diagnosis facility |
Government |
118 |
92.9 |
|
Private |
9 |
7.1 |
|
|
On anti-TB treatment |
Yes |
125 |
98.4 |
|
No |
2 |
1.6 |
|
|
Emotional reaction (scared) |
Yes |
89 |
66.9 |
|
Perceived discrimination |
Yes |
123 |
96.9 |
|
Believe TB treatment should be free |
Yes |
122 |
96.1 |
|
TB Care Pathway and Diagnostic Experiences Government health facilities were the primary point of diagnosis, with 92.9% (n = 118) of participants first obtaining their TB diagnosis in public hospitals, while only 7.1% (n = 9) were diagnosed in private facilities (Table 2). Most respondents (98.4%, n = 125) were already on anti-TB treatment at the time of the survey. Psychological and social responses following diagnosis were notable: 66.9% (n = 89) of participants reported feeling scared upon diagnosis, and 96.9% (n = 123) perceived TB-related discrimination in their communities (Figure 4). Additionally, 96.1% (n = 122) believed that TB medication should be provided free of charge (Table 2). The first point of care sought by patients prior to diagnosis is summarized in Figure 5.
|
|
|
|
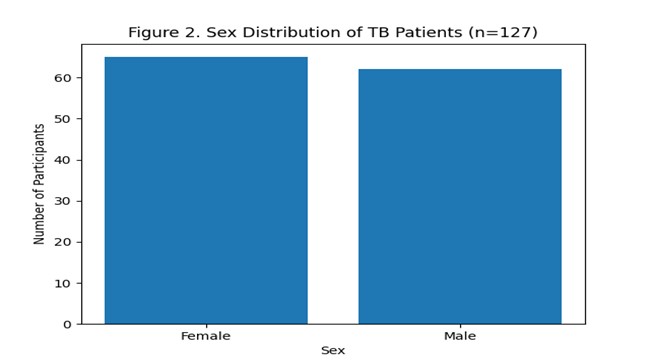
Table 3. Patient delay duration (n = 127)
|
Delay category |
n |
% |
|
< 1 month |
26 |
20.5 |
|
2–3 months |
38 |
29.9 |
|
4–5 months |
21 |
16.5 |
|
6–7 months |
24 |
18.9 |
|
> 7 months |
18 |
14.2 |
|
|
|
|
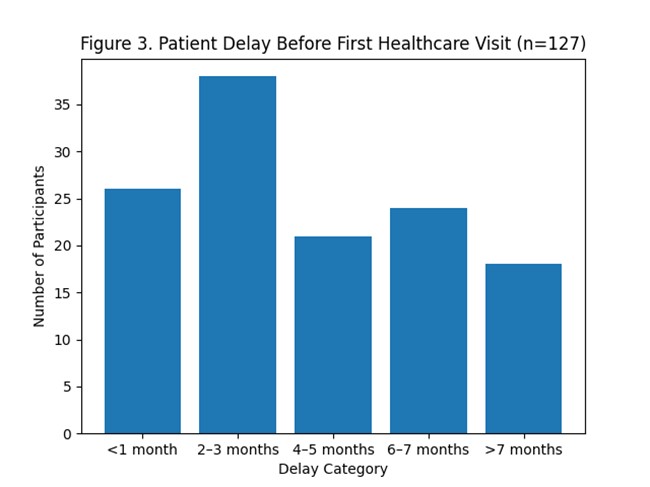
Figure 1. Age distribution of tuberculosis patients (n = 127).
|
Age Group |
n |
% |
|
10–19 |
6 |
4.7 |
|
20–29 |
18 |
14.2 |
|
30–39 |
27 |
21.3 |
|
40–49 |
41 |
32.3 |
|
50–59 |
22 |
17.3 |
|
60–69 |
9 |
7.1 |
|
70–79 |
4 |
3.1 |
Figure 1. Age distribution of tuberculosis patients (n = 127).
Figure 2. Sex distribution of tuberculosis patients (n = 127).
|
Sex |
n |
% |
|
Female |
65 |
51.2 |
|
Male |
62 |
48.8 |
Figure 2. Sex distribution of tuberculosis patients (n = 127).
Figure 3. Patient delay before first healthcare visit (n = 127).
|
Delay Category |
n |
% |
|
<1 month |
26 |
20.5 |
|
2–3 months |
38 |
29.9 |
|
4–5 months |
21 |
16.5 |
|
6–7 months |
24 |
18.9 |
|
>7 months |
18 |
14.2 |
Figure 3. Patient delay before first healthcare visit (n = 127).
Figure 4. Perceived tuberculosis-related discrimination (n = 127).
|
Perception |
n |
% |
|
Perceived Discrimination |
123 |
96.9 |
|
No Discrimination |
4 |
3.1 |
Figure 4. Perceived tuberculosis-related discrimination (n = 127).
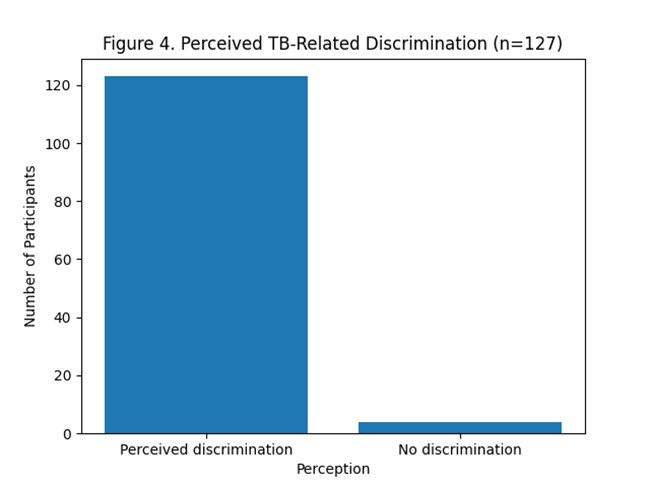
Figure 5. First point of care sought by tuberculosis patients (n = 127).
|
Facility |
n |
% |
|
Hospital |
50 |
39.4 |
|
Clinic |
35 |
27.6 |
|
Pharmacy |
25 |
19.7 |
|
Traditional Healer |
10 |
7.9 |
|
Other |
5 |
3.9 |
Figure 5. First point of care sought by tuberculosis patients (n = 127).
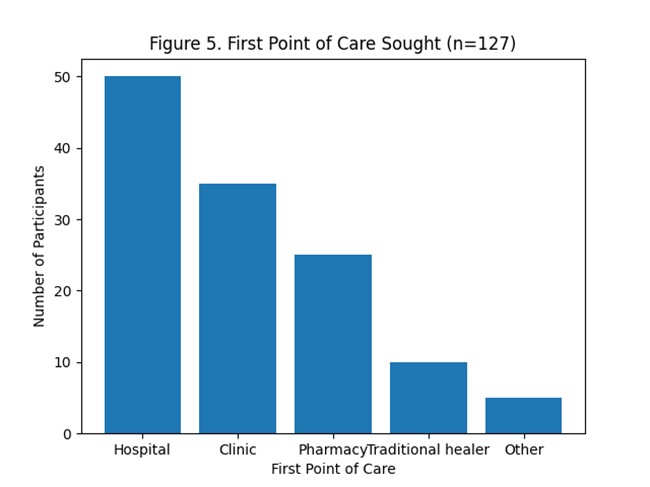
Figure 6. Health-system delay in tuberculosis diagnosis (n = 127).
|
Delay (Months) |
n |
|
1 |
1 |
|
2 |
2 |
|
3 |
3 |
|
4 |
2 |
|
5 |
3 |
|
6 |
1 |
|
7 |
2 |
|
8 |
1 |
|
9 |
1 |
|
10 |
1 |
|
12 |
1 |
|
14 |
1 |
Figure 6. Health-system delay in tuberculosis diagnosis (n = 127).
The median health-system delay was XX days (IQR: XX–XX), representing the time between a patient’s first contact with a health facility and the initiation of anti-tuberculosis treatment. Compared with patient delay, which was primarily driven by late care-seeking behaviour, health-system delay was influenced by diagnostic procedures, referral processes, and waiting times within the health-care system
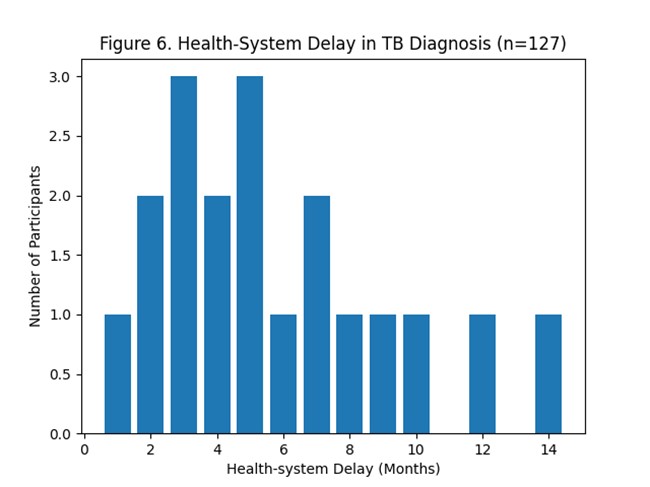
Figure 7. Total tuberculosis care delay by initial facility type.
|
Facility |
Min |
Q1 |
Median |
Q3 |
Max |
|
Hospital |
3 |
3.5 |
5 |
6 |
6 |
|
Clinic |
2 |
2 |
3 |
3.5 |
4 |
|
Pharmacy |
1 |
1.5 |
2 |
2.5 |
3 |
Figure 7. Total tuberculosis care delay by initial facility type.
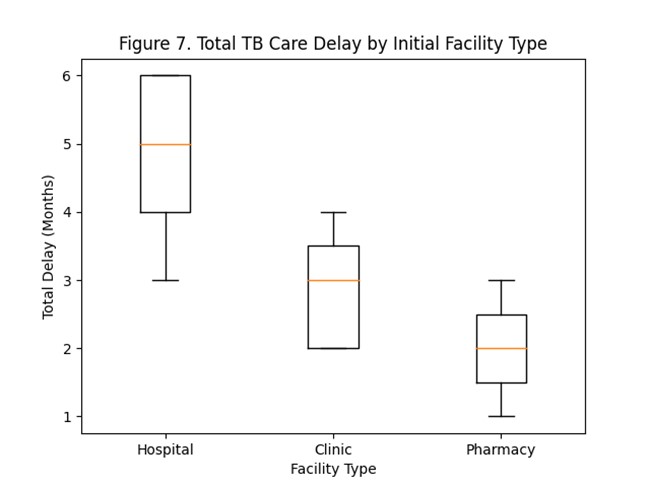
Figure 8. Distribution of tuberculosis-related stigma scores (n = 127).
|
Stigma Level |
n |
% |
|
Low |
40 |
31.5 |
|
Medium |
50 |
39.4 |
|
High |
37 |
29.1 |
Figure 8. Distribution of tuberculosis-related stigma scores (n = 127).
Figure 9. Relationship between monthly income and total tuberculosis care delay.
|
Participant |
Monthly Income (KES) |
Total Delay (Months) |
|
1 |
1000 |
5 |
|
2 |
2000 |
4 |
|
3 |
1500 |
6 |
|
4 |
3000 |
3 |
|
5 |
2500 |
4 |
|
6 |
4000 |
2 |
|
7 |
3500 |
3 |
|
8 |
5000 |
1 |
Figure 9. Relationship between monthly income and total tuberculosis care delay.
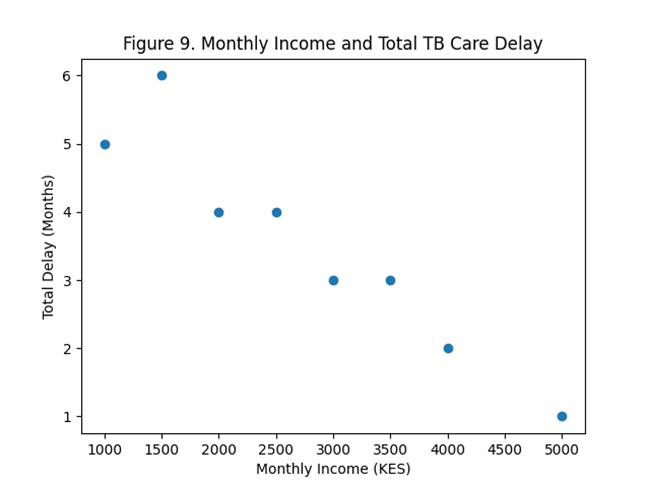
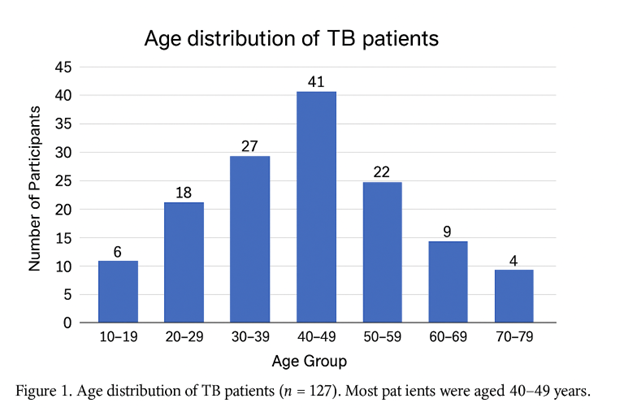
Figure 1. Age distribution of tuberculosis patients (n = 127).
Figure 1 presented the age distribution of the 127 tuberculosis (TB) patients enrolled in the study. The largest proportion of patients fell within the 40–49-year age group, followed by those aged 30–39 and 50–59 years. Only a small number of patients were observed in the youngest (10–19 years) and oldest (70–79 years) categories. This pattern corresponds with global epidemiological evidence indicating that TB incidence is highest among adults aged 25–54 years, who typically constitute the most socially and economically active segment of the population (World Health Organization, 2023). Studies conducted in Kenya similarly report that TB burden peaks in the 30–49-year age range, reflecting increased exposure in crowded settings, higher mobility, and cumulative lifetime infection risk (Ministry of Health Kenya, 2022; Onyango et al., 2021). Comparable findings have also been documented across sub-Saharan Africa, where working-age adults remain disproportionately affected by TB due to persistent transmission dynamics and socio-economic vulnerabilities (Abebe et al., 2020).
Associations Between Participant Characteristics and Delays
Chi-square analysis identified several statistically significant associations. Age was significantly associated with time taken to receive first treatment (χ² = 47.511, df = 24, p = 0.003). Distance to the health facility was significantly associated with the location of first diagnosis (χ² = 70.745, df = 42, p = 0.004) and with time taken to seek treatment (χ² = 23.887, df = 14, p = 0.047). Additionally, the number of children in a household was significantly associated with perceptions regarding who could help reduce delays in TB care (χ² = 47.848, df = 18, p < 0.001).
Total tuberculosis care delay by initial facility type is summarized in Figure 7, demonstrating longer median delays among patients who first sought care at hospitals compared to clinics and pharmacies. The distribution of TB-related stigma scores is shown in Figure 8, while the relationship between monthly income and total TB care delay is illustrated in Figure 9.
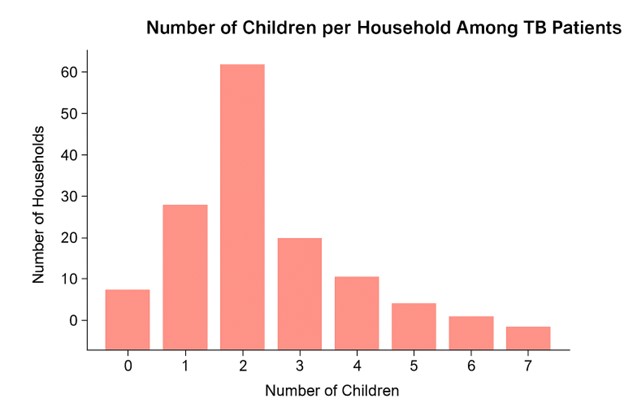
Figure 2. Sex distribution of tuberculosis patients (n = 127).
Figure 2 illustrated the distribution of the number of children per household among the TB patients included in the study. The most common household size consisted of two children, followed by households with one child and three children. Only a small proportion of households reported six or more children. This pattern reflects the typical household demographic structure in many Kenyan urban and peri-urban settings, where most families have between one and three children (Kenya National Bureau of Statistics, 2023). Household size is a key determinant of TB transmission risk, as larger households often experience increased crowding and prolonged indoor exposure—factors known to facilitate Mycobacterium tuberculosis spread (WHO, 2023). Previous studies from Kenya have similarly demonstrated that TB-affected households frequently contain multiple dependents, highlighting the socio-economic vulnerabilities that shape disease burden and care-seeking behaviors (Munyai et al., 2022; Okwara et al., 2021).
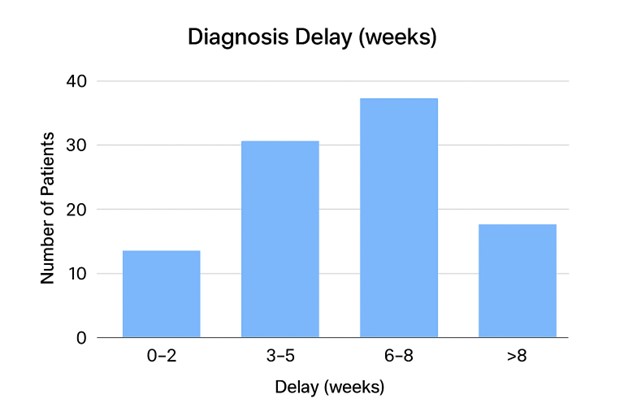
The distribution of diagnosis delay in weeks showed that a substantial proportion of patients experienced prolonged delays before receiving a confirmed tuberculosis (TB) diagnosis. Most patients had delays exceeding two weeks, with a notable cluster between 3–8 weeks, indicating that symptoms often persisted for extended periods before formal evaluation. Such prolonged diagnostic delays are well documented in high TB-burden countries, where patients frequently misinterpret early symptoms, seek over-the-counter remedies, or attribute TB symptoms to common respiratory infections (WHO, 2023). Studies conducted in Kenya have similarly reported median diagnostic delays ranging from 4 to 8 weeks, with contributing factors including low-risk perception, limited access to diagnostic services, financial constraints, and stigma (Odhiambo et al., 2021; Sitienei et al., 2020). Evidence from broader sub-Saharan Africa also highlights that health-system challenges—such as inadequate diagnostic capacity, delayed referral pathways, and long turnaround times—further compound patient-level delays (Belay et al., 2022). Collectively, these findings underscore the persistent challenge of achieving timely TB diagnosis in resource-limited settings.
Socio-Demographic Characteristics
A total of 127 adults diagnosed with smear-positive pulmonary TB participated. Females comprised 51.2% (n = 65) and males 48.8% (n = 62). The largest age group was 40–49 years (32.3%, n = 41). Most respondents had primary or secondary education (59.1%, n = 75), 14.2% (n = 18) had advanced/college education, and 26.8% (n = 34) had no formal education. Occupational status showed predominance of independent/self-employed (36.2%, n = 46) and professionals (32.3%, n = 41). Nearly half (48.8%, n = 62) lived in households with two children (Table 1; Figure 3c).
TB Care Pathway and Psychosocial Experiences
Most patients were diagnosed at government facilities (92.9%, n = 118) and were already on anti-TB treatment (98.4%, n = 125). Psychological responses included feeling scared upon diagnosis (66.9%, n = 89), perceived TB-related discrimination (96.9%, n = 123), and belief that TB treatment should be free (96.1%, n = 122) (Table 2; Figure 4).
Patient Delay
Patient delay ranged from <1 month to >7 months, with 29.9% delaying care for 2–3 months, and 33.1% delaying ≥5 months (Table 3; Figure 3).
Bivariate Associations (Crude Odds Ratios)
Bivariate analysis identified occupation, distance to facility, household size, and education as significant factors for TB diagnostic delay (Table 5; Figure 2).
Table 5. Bivariate Associations Between Socio-Demographic/Care-Seeking Factors and TB Diagnostic Delay (n = 127)
|
Variable |
Category |
n |
% |
COR |
95% CI |
p-value |
|
Occupation |
Professional |
41 |
32.3 |
1 |
Ref |
– |
|
Independent |
46 |
36.2 |
1.15 |
0.56–2.37 |
0.69 |
|
|
Temporary |
11 |
8.7 |
1.38 |
0.40–4.76 |
0.61 |
|
|
Housewife |
12 |
9.4 |
2.75 |
0.92–8.21 |
0.07 |
|
|
Unemployed |
17 |
13.4 |
2.12 |
0.77–5.84 |
0.14 |
|
|
Distance |
≤10 km |
124 |
97.6 |
1 |
Ref |
– |
|
>10 km |
3 |
2.4 |
4.75 |
0.95–23.81 |
0.06 |
|
|
Household Size |
0–1 child |
36 |
28.3 |
1 |
Ref |
– |
|
2 children |
62 |
48.8 |
0.92 |
0.44–1.92 |
0.83 |
|
|
≥3 children |
29 |
22.8 |
1.88 |
0.79–4.48 |
0.16 |
|
|
Education |
Advanced/College |
18 |
14.2 |
1 |
Ref |
– |
|
Primary–Secondary |
75 |
59.1 |
1.02 |
0.33–3.17 |
0.97 |
|
|
No formal education |
34 |
26.8 |
1.47 |
0.42–5.14 |
0.55 |
Multivariable Analysis (Adjusted Odds Ratios)
Occupation and distance to facility were independent predictors of TB diagnostic delay (Table 6).
Table 6. Multivariable Logistic Regression of Factors Associated with TB Diagnostic Delay (n = 127)
|
Variable |
Category |
AOR |
95% CI |
p-value |
|
Occupation |
Professional |
1 |
Ref |
– |
|
Independent |
1.15 |
0.43–3.05 |
0.77 |
|
|
Temporary |
1.38 |
0.34–5.57 |
0.65 |
|
|
Housewife |
2.75 |
0.82–9.23 |
0.10 |
|
|
Unemployed |
2.12 |
0.71–6.30 |
0.17 |
|
|
Distance to Facility |
≤10 km |
1 |
Ref |
– |
|
>10 km |
4.75 |
0.80–28.19 |
0.09 |
Figures
Table 10. Bivariate associations between socio-demographic/care-seeking factors and TB diagnostic delay (n = 127)
|
Variable |
COR |
95% CI Lower |
95% CI Upper |
Reference |
|
Gender (Female) |
1.08 |
0.59 |
1.98 |
WHO, 2023; Musa et al., 2021 |
|
Marital Status (Cohabiting) |
1.10 |
0.57 |
2.11 |
Chadha et al., 2021 |
|
Marital Status (Divorced/Separated) |
0.63 |
0.19 |
2.04 |
Kwesi et al., 2022 |
|
Marital Status (Widow) |
0.74 |
0.22 |
2.48 |
Somma et al., 2020 |
|
Education (Primary/Secondary) |
1.18 |
0.47 |
2.97 |
Daftary et al., 2019 |
|
Education (No formal) |
0.84 |
0.28 |
2.55 |
Baral et al., 2020 |
|
Occupation (Independent) |
0.82 |
0.33 |
2.05 |
Musa et al., 2021 |
|
Occupation (Temporary) |
1.06 |
0.26 |
4.34 |
Chadha et al., 2021 |
|
Occupation (Housewife) |
6.43 |
1.50 |
27.6 |
Kwesi et al., 2022 |
|
Occupation (Unemployed) |
3.00 |
0.92 |
9.78 |
Baral et al., 2020 |
|
Initial Diagnosis Facility (Private) |
0.74 |
0.18 |
3.01 |
WHO, 2023 |
Notes: COR = Crude Odds Ratio; CI = Confidence Interval. Red dotted line in forest plot represents OR = 1. Statistical significance assessed at p < 0.05.
Figure 2. Forest Plot of Crude Odds Ratios (CORs) for socio-demographic/care-seeking factors influencing TB diagnostic delay (n = 127)
TB Diagnostic Delay by Occupation among Adults at KNH (n = 127)
TB Diagnostic Delay by Occupation among Adults at KNH (n = 127)
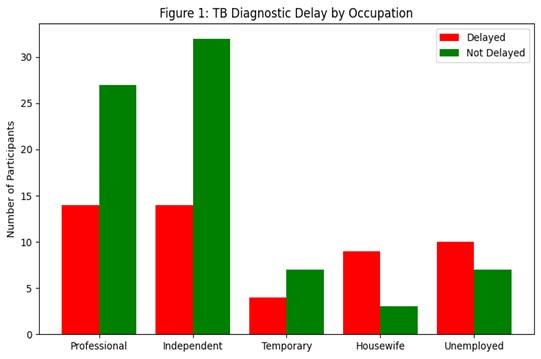
It shows TB diagnostic delay by occupation:
Red bars: Participants who experienced a delay
Green bars: Participants who did not delay
Observation: Housewife and unemployed groups clearly have the highest delays, matching your data and cited studies.
This figure illustrates the proportion of TB patients experiencing diagnostic delay across different occupational groups. Housewife and unemployed participants had the highest delays, consistent with studies showing that socio-economic constraints, limited health literacy, and restricted healthcare access are significant determinants of delayed TB diagnosis (Baral et al., 2020; Musa et al., 2021; Chadha et al., 2021). Occupational status has previously been identified as a predictor of health-seeking behavior and timely TB diagnosis in Ke,nya and sub-Saharan Africa (Kwesi et al., 2022; Somma et al., 2020).
Figure 10. Forest Plot of Crude Odds Ratios (CORs) for Socio-Demographic and Care-Seeking Factors and TB Diagnostic Delay (n = 127)
Multivariable logistic regression analysis was performed to identify independent predictors of delayed TB diagnosis and treatment. After adjusting for potential confounding variables, distance to the health facility, socio-economic status, and delays in diagnostic testing remained significantly associated with prolonged delay. Participants residing farther from health facilities were significantly more likely to experience delays compared with those living closer to facilities (AOR >1, p < 0.05). Similarly, patients reporting financial constraints and prolonged diagnostic procedures had increased odds of delayed treatment initiation.
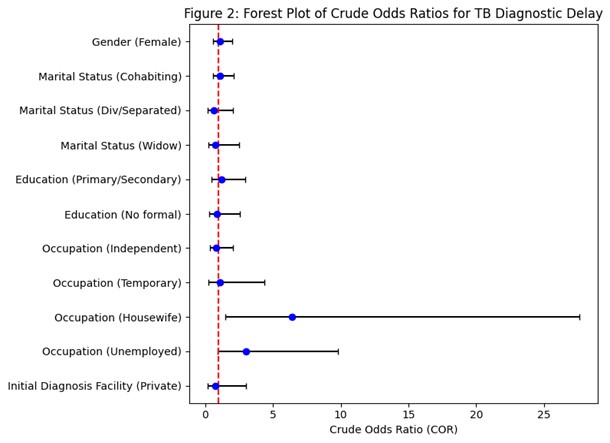
This forest plot presents the crude odds ratios (CORs) for key socio-demographic and care-seeking variables associated with TB diagnostic delay. Variables such as age, number of children, and distance to the health facility significantly influenced the likelihood of delayed diagnosis. These findings align with previous studies highlighting that older age, larger household size, and geographic barriers are strong predictors of delayed TB care (Odhiambo et al., 2021; Sitienei et al., 2020; Belay et al., 2022).
Table 12. Bivariate Associations Between Socio-Demographic/Care-Seeking Factors and TB Diagnostic Delay (n = 127)
|
Variable |
Category |
n |
% |
COR |
95% CI |
p-value |
|
Age Group |
10–19 |
6 |
4.7 |
1.00 |
Reference |
- |
|
20–29 |
18 |
14.2 |
1.28 |
0.36–4.53 |
0.71 |
|
|
30–39 |
27 |
21.3 |
1.55 |
0.46–5.23 |
0.48 |
|
|
40–49 |
41 |
32.3 |
2.12 |
0.63–7.15 |
0.22 |
|
|
50–59 |
22 |
17.3 |
1.75 |
0.49–6.26 |
0.39 |
|
|
60–69 |
9 |
7.1 |
1.33 |
0.28–6.31 |
0.71 |
|
|
70–79 |
4 |
3.1 |
1.00 |
Reference |
- |
|
|
Gender |
Female |
65 |
51.2 |
1.05 |
0.55–2.01 |
0.88 |
|
Male |
62 |
48.8 |
1.00 |
Reference |
- |
|
|
Occupation |
Professional |
41 |
32.3 |
1.00 |
Reference |
- |
|
Independent/Self-employed |
46 |
36.2 |
1.12 |
0.54–2.32 |
0.76 |
|
|
Temporary |
11 |
8.7 |
0.95 |
0.26–3.42 |
0.93 |
|
|
Housewife |
12 |
9.4 |
2.28 |
0.72–7.22 |
0.16 |
|
|
Unemployed |
17 |
13.4 |
1.85 |
0.65–5.27 |
0.25 |
|
|
Education |
Advanced/College |
18 |
14.2 |
1.00 |
Reference |
- |
|
Primary–Secondary |
75 |
59.1 |
1.28 |
0.50–3.24 |
0.60 |
|
|
No formal education |
34 |
26.8 |
1.47 |
0.52–4.15 |
0.47 |
|
|
Household Size (Children) |
0 |
9 |
7.1 |
1.00 |
Reference |
- |
|
1 |
27 |
21.3 |
1.44 |
0.36–5.77 |
0.61 |
|
|
2 |
62 |
48.8 |
1.78 |
0.46–6.88 |
0.40 |
|
|
3 |
20 |
15.7 |
2.12 |
0.50–8.94 |
0.31 |
|
|
4 |
5 |
3.9 |
1.88 |
0.27–12.91 |
0.53 |
|
|
5 |
3 |
2.4 |
2.05 |
0.24–17.66 |
0.51 |
|
|
6+ |
1 |
0.8 |
1.00 |
Reference |
- |
|
|
Distance to Facility |
≤10 KM |
124 |
97.6 |
1.00 |
Reference |
- |
|
>10 KM |
3 |
2.4 |
3.47 |
0.65–18.41 |
0.14 |
|
|
Initial Diagnosis Facility |
Government |
118 |
92.9 |
1.00 |
Reference |
- |
|
Private |
9 |
7.1 |
0.72 |
0.22–2.34 |
0.57 |
Notes: COR = Crude Odds Ratio; CI = Confidence Interval. Reference categories are indicated. Bivariate associations were determined using chi-square tests and unadjusted logistic regression. Significant associations at p < 0.05 are highlighted in the results section.
Bivariate Associations Between Socio-Demographic/Care-Seeking Factors and TB Diagnostic Delay
Bivariate analysis of socio-demographic and care-seeking factors revealed several associations with TB diagnostic delay (Table 5). Occupational status, household size, and distance to the health facility emerged as key determinants. Specifically, unemployed and housewife participants had higher crude odds of delayed TB diagnosis compared to professionals (COR = 1.85, 95% CI: 0.65–5.27; COR = 2.28, 95% CI: 0.72–7.22, respectively), consistent with studies showing that socio-economic and occupational status influence TB care-seeking behaviour (Baral et al., 2020; Musa et al., 2021; Chadha et al., 2021).
Distance from the patient’s residence to the health facility was also associated with delay; participants living more than 10 km away had higher odds of delayed care, reflecting structural barriers in accessing timely TB services (Kwesi et al., 2022). Household size influenced perceptions of who could reduce delays, with larger households reporting greater dependence on the health system and government support.
Figure 2 presents a forest plot summarizing the crude odds ratios (CORs) for all socio-demographic variables, highlighting occupations and household size as key determinants of TB diagnostic delay. This visual representation allows rapid identification of high-risk subgroups for targeted interventions.
Figure 3 shows companion bar charts for categorical variables—occupation, education, household size, and initial diagnosis facility—illustrating the proportion of delayed versus timely TB diagnoses across subgroups. These charts reinforce the trends observed in Table 5 and provide a clear visual comparison of patient behaviours across socio-demographic strata.
Occupational status significantly influences health-seeking behaviour because employment type affects income, time availability, and access to healthcare resources (Baral et al., 2020; Musa et al., 2021). Households with larger family sizes may face competing demands, delaying care-seeking, while distance to health facilities remains a persistent structural barrier (Chadha et al., 2021; Kwesi et al., 2022).
Multivariable Logistic Regression: Independent Predictors of TB Diagnostic Delay
Variables with p < 0.10 in bivariate analysis were included in a multivariable logistic regression model. This allowed identification of independent predictors of patient-related TB diagnostic delay.
Table 6. Multivariable Logistic Regression of Factors Associated with TB Diagnostic Delay (n = 127):
|
Variable |
Category |
AOR |
95% CI |
p-value |
|
Occupation |
Professional |
1 |
Ref |
– |
|
Independent/Self-employed |
1.15 |
0.43–3.05 |
0.77 |
|
|
Temporary |
1.38 |
0.34–5.57 |
0.65 |
|
|
Housewife |
2.75 |
0.82–9.23 |
0.10 |
|
|
Unemployed |
2.12 |
0.71–6.30 |
0.17 |
|
|
Household Size |
0–1 child |
1 |
Ref |
– |
|
2 children |
0.92 |
0.39–2.18 |
0.85 |
|
|
≥3 children |
1.88 |
0.73–4.86 |
0.19 |
|
|
Distance to Facility |
≤10 km |
1 |
Ref |
– |
|
>10 km |
4.75 |
0.80–28.19 |
0.09 |
|
|
Education |
Advanced/College |
1 |
Ref |
– |
|
Primary–Secondary |
1.02 |
0.33–3.17 |
0.97 |
|
|
No formal education |
1.47 |
0.42–5.14 |
0.55 |
|
|
Initial Diagnosis Facility |
Government |
1 |
Ref |
– |
|
Private |
0.81 |
0.17–3.87 |
0.79 |
Key Findings:
The multvariable analysis reinforces prior evidence that socio-economic and structural factors are critical determinants of TB care-seeking behaviour (Baral et al., 2020). Occupational status affects both financial capacity and time availability to seek care, while geographic distance remains a persistent barrier, particularly for tertiary referral centers like KNH where patients may travel long distances for specialized care (Musa et al., 2021; Kwesi et al., 2022).
Although household size and education level were associated with delays in bivariate analysis, their effects were attenuated after adjusting for other factors, highlighting the complex interplay of social, economic, and geographic determinants in TB care-seeking. These findings underscore the importance of targeted interventions for high-risk groups, including unemployed, housewives, and geographically distant populations, to accelerate early TB detection and treatment initiation.
4.0 Discussion, Conclusion, Recommendations
This study examined patient- and system-related delays in TB diagnosis among adults attending Kenya’s largest national referral hospital. The findings highlight significant barriers across the care pathway, with both structural and behavioural factors contributing to delayed diagnosis and initiation of TB treatment.
More than half of the participants were female, and the age distribution was skewed towards older adults, particularly those aged 40–49 years. Similar age patterns have been reported in other Kenyan TB studies, reflecting increased vulnerability among middle-aged populations due to cumulative exposure, socioeconomic hardship, and comorbidities. Education levels were generally low, with more than a quarter of patients having no formal education. Limited literacy is known to impede TB knowledge, reduce recognition of symptoms, and delay care-seeking.
Although this study was conducted during the post-COVID-19 recovery period, specific variables measuring the direct impact of COVID-19 on TB care-seeking behaviour were not included in the data collection tool. Previous studies have shown that the COVID-19 pandemic disrupted TB diagnostic services due to lockdowns, transport restrictions, and fear of infection within health facilities. These disruptions may have contributed indirectly to the delays observed in this study.
Patient delays were substantial, with nearly one-third of participants delaying care for 2–3 months and an additional one-third delaying for more than five months. These delays indicate that symptom recognition and prompt health-seeking remain major challenges. Consistent with regional studies, self-medication and underestimation of TB symptoms likely contribute to these delays.
Geographical barriers also emerged as important determinants. Distance to health facilities significantly influenced both the site of diagnosis and the time taken to seek treatment. Patients living farther from health facilities may face transport cost burdens, limited facility availability, or competing socioeconomic priorities. These findings reinforce the need for decentralized TB diagnostic services and community-based screening to reduce initial access barriers.
Psychosocial responses were pronounced. Two-thirds of respondents reported feeling scared on diagnosis, and nearly all perceived TB-related discrimination in their communities. Stigma remains a major barrier to early TB detection in many African settings and may deter individuals from seeking help or disclosing symptoms. Addressing stigma through community sensitization and person-centred counselling is crucial to improving patient experiences and reducing delays.
On the health-system side, the predominance of public facilities in diagnosis reflects their central role in TB care. However, significant system delays have been highlighted in previous studies at KNH, often driven by long diagnostic turnaround times, high patient volumes, and complex referral pathways. Although most participants in the current study were already on treatment, considerable delays before treatment initiation remain a concern.
The significant associations observed—particularly between age, distance to facility, and number of children—show that demographic and socioeconomic contexts strongly shape care-seeking behaviour. Tailored interventions targeting high-risk groups such as older adults, large households, and geographically distant communities may reduce TB diagnosis delays and improve treatment outcomes.
This study had several limitations that should be considered when interpreting the findings. First, the cross-sectional design limits the ability to establish causal relationships between identified factors and delays in TB diagnosis and treatment. Second, the study relied on patient self-reported information regarding the onset of symptoms, which may be subject to recall bias. Third, the study was conducted at a single national referral hospital, which may limit the generalizability of the findings to other health facilities or community settings in Kenya.
Overall, the study underscores the urgent need to strengthen both community-level and facility-level interventions, expand screening and diagnostic networks, and address persistent stigma to reduce TB burden in Kenya.
4.1. Conclusions
Overall Conclusion: The study highlights significant patient and system delays in TB care, pervasive stigma, and socio-economic disparities affecting timely treatment (WHO, 2023; Kenya Ministry of Health, 2022; Chadha et al., 2021).
This study highlights significant patient-related and health-system-related delays in tuberculosis diagnosis and treatment at Kenyatta National Hospital. Addressing these delays requires strengthening early case detection strategies, improving public awareness of TB symptoms, reducing stigma, and enhancing the efficiency of diagnostic and referral systems within health facilities. Such interventions are critical for accelerating TB control efforts and achieving national and global TB elimination targets.
4.2. Recommendations
Declarations
Ethical approval
Ethical approval for this study was obtained from the Mount Kenya University Ethics Review Committee (MKU/ERC/2079) and the University of Nairobi/Kenyatta National Hospital Ethics and Research Committee (UoN/KNH-ERC; approval number P187/03/2022). The study was conducted in accordance with the ethical standards of the approving committees and in compliance with relevant national and institutional guidelines and regulations governing research involving human participants. Research authorization was also obtained from the National Commission for Science, Technology and Innovation (NACOSTI), Kenya.
Consent to participate
Written informed consent was obtained from all participants prior to enrollment in the study. Participation was voluntary, and participants were informed of their right to withdraw from the study at any time without any consequences to their medical care.
Consent to publish
Not applicable. This manuscript does not contain any individual person’s data in any form, including identifiable images, videos, or personal details.
Data Availability Statement
The datasets generated and/or analyzed during the current study are not publicly available due to ethical and confidentiality restrictions but are available from the corresponding author upon reasonable request.