International Journal of Epidemiology And Public Health Research
OPEN ACCESS | Volume 9 - Issue 1 - 2026
ISSN No: 2836-2810 | Journal DOI: 10.61148/2836-2810/IJEPHR
Tah Aldof Yoah1,2*, Tendongfor Nicholas1, Njukang Ernest Nkem1, Kah Emmanuel Nji1, Njoh Josephine3, Nde Fon Peter1, Enow Orock George4, Anna Longdoh Njunda5
¹1Department of Public Health and Hygiene, Faculty of Health sciences, University of Buea, Cameroon.
2Department of Nursing, School of Health Sciences, Biaka University Institute of Buea, Cameroon.
3Department of Paediatric HIV, Baptist Hospital Mutengene, Cameroon.
4Department of Biomedical Sciences, Faculty of Health sciences, University of Buea, Cameroon.
5Department of Medical Microbiology and Parasitology, Faculty of Health sciences, University of Buea, Cameroon.
*Corresponding author: Tah Aldof Yoah, Department of Public Health and Hygiene, Faculty of Health sciences, University of Buea, Cameroon.
Received: March 10, 2026 | Accepted: March 22, 2026 | Published: April 06, 2026
Citation: Tah A Yoah, Nicholas T, Njukang E Nkem, Kah E Nji, Josephine N, Nde F Peter, Enow O George, Anna L Njunda., (2026) “Prevalence and Associated Risk Factors of Non-Communicable Diseases Among HIV Patients on Antiretroviral Therapy in Fako Division”. International Journal of Epidemiology and Public Health Research, 9(2); DOI: 10.61148/2836-2810/IJEPHR/191.
Copyright: © 2026. Tah Aldof Yoah. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Background: Non-communicable diseases (NCDs) have reached epidemic proportion among people living with HIV (PLHIV) and this could have a negative bearing on the quality of life and survival of these patients. The prevalence of NCDs remain largely unquantified in the context of the universal test and treat (UTT) approach. Aim: The aim of this study was to determine the prevalence and associated risk factors of NCDs among HIV patients on antiretroviral therapy (ART) in Fako Division, South West Region of Cameroon. Methodology: A cross-sectional survey conducted in the months of June to August 2021. Patients’ files alongside consultation by experience nurses were used to identify NCDs. A total of 1440 HIV patients were sample from 5 HTCs selected purposively (high number of patients receiving treatment in these Centers). A serial or consecutive sampling technique was used to recruit participants in the HTCs. The NCDs included in this study were hypertension, diabetes mellitus, osteoporosis, renal impairment, cardiomyopathy and asthma. Data was entered in SPSS 25 and analyzed using stata 13. Multivariate logistic regression was used to identify independent determinants. Statistical significance was set at p<0.05. Results: The mean age of the participants was 45.6 (SD = 7.3) years. Of 1440 participants sampled, 1023 (71.0%) were females, 753 (52.4%) were either married or cohabiting and 156 (10.9%) had no formal education. The overall prevalence of NCDs was 26.9% [95%CI: 24.6-29.2] (hypertension, 18.1% [95%CI: 16.2-20.2]; osteoporosis, 8.7% [95%CI: 7.3-10.5]; diabetes mellitus, 4.8% [95%CI: 3.6-6.0]; renal impairment, 1.9% [95%CI: 1.3-2.7]; asthma, 0.4% [95%CI: 0.1-0.9] and cardiomyopathy, 1.1% [95%CI: 0.6-1.7]). The prevalence of multi-morbidity was 7.7% (95% CI: 6.4 – 9.2). Age > 51years (AOR 3.90; 95% CI 1.50-10.11), family history of NCDs (AOR 5.40; 95% CI 3.69-7.54), smoking (AOR 3.51; 95% CI 1.96-6.11), WHO HIV clinical stage IV (AOR 4.10; 95 % CI 2.99-5.96) and duration on ART for more than 5 years (AOR 2.03; 95% CI 1.39-2.99) were significant predictors of NCDs among HIV patients. Conclusion: The prevalence of NCDs was high. Socio-demographic factors, clinical factors and duration on ART significantly predicted the occurrence of NCDs. Recommendation: HTCs in Fako should routinely and actively screen for NCDs.
HIV, Prevalence, determinants, Non-communicable diseases, ART, UTT, Fako Division
The burden of non-communicable diseases (NCD) and their modifiable risk factors is on the rise in Sub-Saharan Africa (SSA). Among people living with HIV (PLHIV), epidemiological studies have revealed a trend of increasing prevalence of four major risk factors of NCDs; hypertension, hyperglycemia, dyslipidemia and obesity [1-3]. With the increasing availability of antiretroviral therapy (ART), the ageing HIV population is susceptible to traditional risk factors for NCDs. Furthermore, both the HIV virus and prolonged ART use have been associated with dyslipidemia, insulin resistance, and atherosclerosis, interacting with traditional risk factors to increase the risk of NCDs among PLHIV [3, 4]. The increased NCDs risk among PLWH has the potential to threaten the success of ART use, causing morbidity, poor quality of life and premature mortality.
In Cameroon, the universal test and treat approach was instituted in 2016. This novel approach requires placing all persons testing positive for HIV on ART irrespective of their immunological and clinical statuses. Since the institution of this novel strategy in Cameroon, very few studies have investigated the epidemiology of NCDs among PLHIV in the context of the universal test and treat strategy. Approximately more than 35 million deaths are caused by non-communicable diseases (NCDs) on an annual basis. Morbidity and mortality due to NCDs contribute significant threat globally on health and economy of individuals, societies and health systems [5, 6]. The four main NCDs which are being targeted for control globally are cardiovascular diseases (CVDs), chronic respiratory diseases, cancers and diabetes and the selected NCD risk factors also targeted for control are tobacco use, harmful alcohol use, salt intake, obesity, raised blood pressure, raised blood glucose and diabetes, and physical inactivity [6].
Coupled to the rapid urbanization observed in Fako Division, the double crises (socio-political and COVID-19 pandemics) could increase the prevalence of NCDs through either inaccessibility to health facilities and/or stressors. The lock-downs imposed by these two crises have led to increase in sedentary life, psychological stress and inadequate exercise with a long term consequence of spikes in NCDs such as hypertension and diabetes [7]. These double crises have considerably disrupted the health system through reduction in access to health facilities and reduction in staff and essential medication for patients suffering from NCDs. There is consistent evidence across the globe that the COVID-19 pandemic has considerably disrupted NCDs services. This disruption has been particularly problematic for people living with chronic conditions and requiring long term care. Triage and telemedicine have been used to address this disruption in NCD services across the world.
The prevalence of HIV in Cameroon was 3.9% in 2015 [8]. The World Health Organization (WHO) estimates for deaths attributed to Non-Communicable Diseases (NCD) in Cameroon was 35.0% in 2018 [9]. The prevalence of HIV is 3.6% and that of hypertension is 31.1% in the South West Region (SWR) of Cameroon [2, 8]. The prevalence of hypertension (38.0%) and dyslipidemia (51.0%) in HIV patients on ART have reached epidemic levels in Fako Division, SWR of Cameroon [2, 3]. Fako Division as well as the other fast urbanizing divisions in Cameroon is experiencing a double burden of HIV and NCDs. Although evidence from high-income countries is definitive as to the emerging importance of NCDs in PLHIV, there are far fewer data and research advances regarding such conditions in low and middle income countries (LMICs). In Cameroon, the burden of NCDs and their risk factors among PLHIV remains largely unquantified. This study aimed to describe the prevalence of hypertension, osteoporosis, diabetes mellitus, renal impairment, asthma, cardiomyopathy and multi-morbidity, among HIV positive patients receiving ART in Fako Division.
MATERIALS AND METHODS
Ethical and administrative consideration
For administrative approval, the study protocol was read and approved by South West Regional Delegation of Public Health and heads of the health facilities hosting the HTCs. Ethical clearance was obtained from the Faculty of Health Science Institutional Review Board, University of Buea, Cameroon
Study design
This was a cross-sectional survey that took place in the months of June to August 2021.
Study area and setting
The study was carried out in in 5 purposively (HTCs receiving the highest number of patients) in Fako Division (Figure 1). These hospitals are specialized HIV treatment centers that provide free antiretroviral therapy in addition to HIV counseling and testing services. According to the RTG of the Regional Delegation of Public Health for the South West Region report for 2020, a total of 18,000 HIV patients were receiving treatment in Fako Division [10]. Patients report every 1, 3 and 6 months for antiretroviral therapy refill depending on the availability of drugs. The number of HIV patients on ART has seen a hick because most patients have been transferred-in from the highly hit conflict areas to Fako Division which is relatively safer. The patients who attend the facility are of different socioeconomic statuses. Information on NCDs is not collected in routine care at the facility during patient visits. Hypertension is sometimes screened for at every clinic visit, diabetes mellitus II, asthma, cardiomyopathy and osteoporosis are usually screened for when a patient complains of certain symptoms during the routine follow up visits. Renal impairment is screened for among patients upon being started on tenofovir based regimen. However, some NCDs are screened for as a result of participation in research projects that require screening for certain ailments / medical conditions.
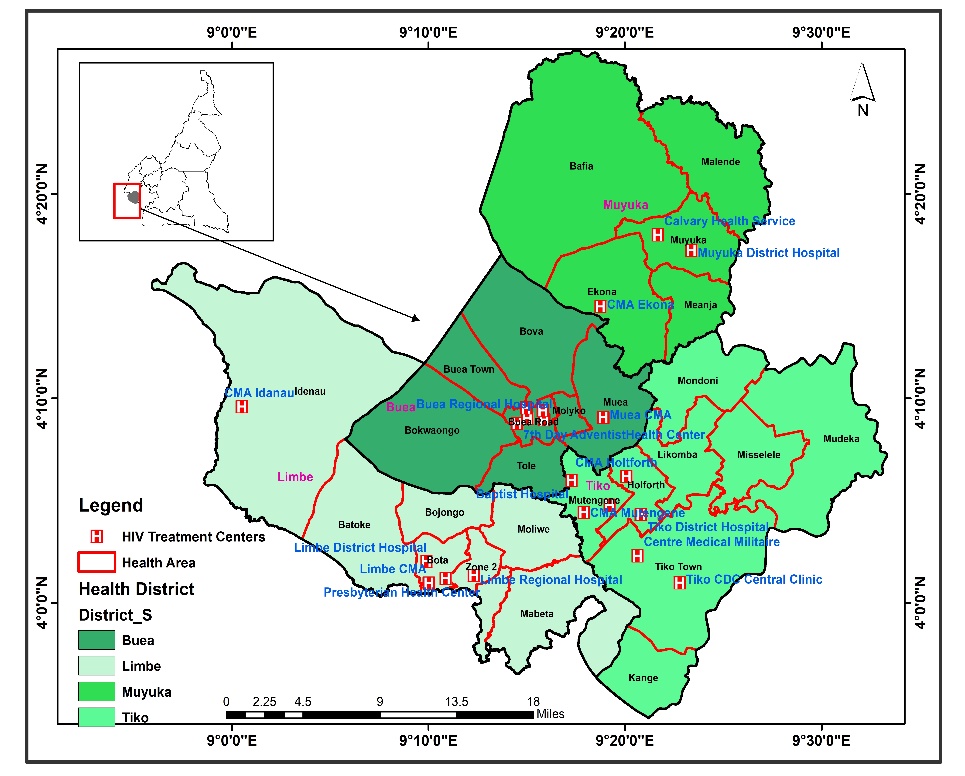
Figure 1: Map showing the HTCs in Fako Division
Study population
The study population consisted of HIV patients aged 18 years and above receiving ART in the selected treatment centers in Fako Division.
Inclusion criteria
Exclusion criteria
Sample size
The single population proportion formula by Fischer for cross-sectional studies was used for the WHO STEP approach [11]. To adjust for the design effect of the sample design, we multiplied the sample size by the design effect. To adjust for anticipated non-response, we divided by the anticipated response rate. Hence, Z-score=1.96; Proportion=38% [2]; marginal error=0.05; Design effect=1.5 and non-response rate=10%.
n = Z2 *p*(1-p)e2*1.5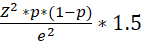
n = (1.96)2*0.38*0.62(0.05)2*1.5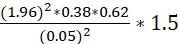
n = 545
To adjust for anticipated non-response, we added 10% of the sample. Thus a minimum total of 600 participants were anticipated and a total of 1440 clients were sampled using probability proportionate to size (PPS) as shown in table 1 below.
Table 1: Proportionate samples for each HTC
|
HIV treatment centre |
Total number of patients |
Number of patients to be sampled |
|
Hospital – 1 |
3972 |
414 |
|
Hospital – 2 |
2738 |
286 |
|
Hospital – 3 |
1026 |
107 |
|
Hospital – 4 |
2042 |
213 |
|
Hospital – 5 |
4028 |
420 |
|
Total |
13,806 |
1440 |
Sampling method
A two stage sampling method was used. In the first stage, a purposeful sampling technique was used to select 5 HIV treatment centers in Fako Division. These treatment centers receive the largest number of clients and relatively secured in the current socio-political context. In the second stage, 1440 HIV patients on ART were recruited from the 5 selected treatment centers based on probability proportionate to size (Figure 2). A serial or consecutive sampling technique was used to recruit participants from the treatment centers until the sample size is reached.
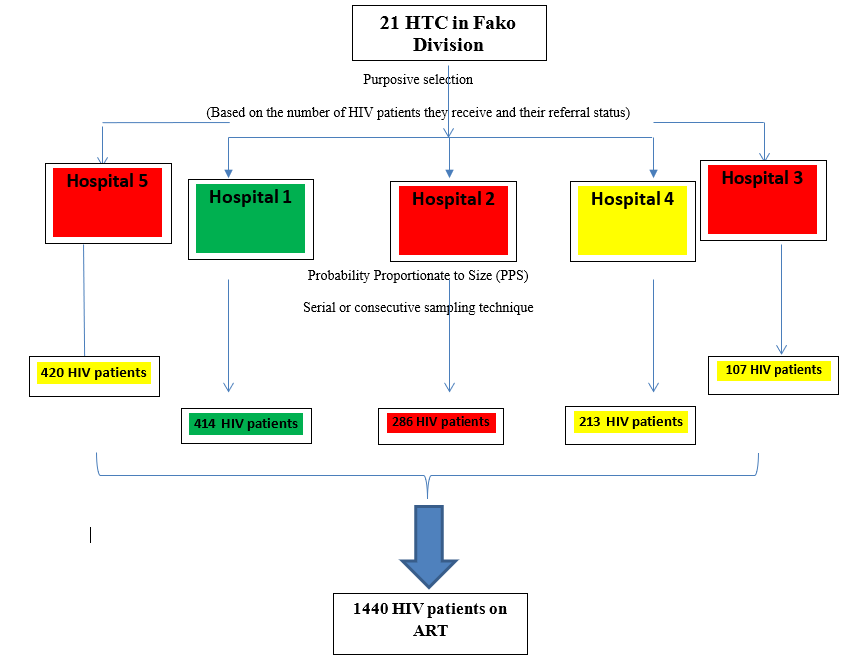
Figure 2: Sampling method for the first cross-sectional study
Study variables
Outcomes of interest: Hypertension, osteoporosis, diabetes mellitus, renal impairment, asthma, cardiomyopathy and multi-morbidity were the outcomes of interest. They were all treated as binary variables. An individual was categorized as having a condition or not.
Other variables: Demographic factors: age, sex, marital status, level of education, occupation
Other health related factors: WHO HIV stage, duration on ART.
Life style factors: smoking, alcohol use.
Data collection
Research experienced personnel with bachelors in nursing and midwifery, already working within the HIV treatment centers were recruited and trained as research assistants. They were responsible for the data extraction and interviews. Patients presenting to the study facility for clinical review and/or drug re-supply, were consented into the study. Relevant information was then extracted from their patient files and entered into a data extraction form by the nurses. The patient files included information on a particular individual from when they were enrolled as patients in the selected HIV treatment centers. At the facility the following criterion is used to determine whether an individual has one of the above diseases:
Hypertension: Individual having a systolic blood pressure (SBP) of 140mmHg or more, or a diastolic blood pressure (DBP) of 90 mm Hg or more, or taking anti-hypertensive medication.
Diabetes mellitus: Having a fasting blood sugar equal to or more than 140mg/dl (7.8mmol/l) on 2 or more occasions, or a Random blood sugar of more than 200mg/dl (11.8mmol/l) or having a history of diagnosis of diabetes mellitus.
Renal impairment: Having urea levels, creatinine levels or potassium levels above the normal range or a calculated glomerular filtration rate (eGFR) of less than 60ml/ min.
Asthma: This is assessed using medical history and proof of asthma relief medicines for example salbutamol inhaler and medical documents showing asthma treatment, clinical examination to confirm presence of bronchi, spirometry to assess reversible airway obstruction with an FEV1 /FVC ratio less than 0.7 in an attack but with marked improvement on administration of bronchodilators that it: an increase in FEV1 (or FVC) of ≥12% and an absolute increase in FEV1 (or FVC) of ≥200mL.
Cardiomyopathy: A chest X-ray indicative of an enlarged heart; or a cardiac echo with a cardiologist report confirming a cardiomyopathy.
Osteoporosis: This is based on a clinical history of low velocity, atraumatic fractures or fragility fractures (hip, vertebrae, spine, forearm and wrist) and associated bone pain, taking medications for bone health, clinical examination and X-ray reports.
Data management
All study data was checked for accuracy, completeness and consistency at the end of each day by the PI, and any identified errors were corrected in real time. Hard copies of the questionnaires were stored in a water proof box file and electronic data bases were kept in password protected computers. An external hard drive was used to back up the data. Double data entry was done using Epi-info version 7.
Data analysis
All data analysis was done using STATA version 13.0 (STATA, College Station, Texas, USA). All continuous variables were summarized using means and standard deviations or medians and inter-quartile ranges, while categorical variables were summarized using proportions or percentages. Prevalence was reported with 95% confidence intervals. Multivariate logistic regression was used to identify the independent risk factors of NCDs among HIV patients on ART in Fako Division, SWR of Cameroon.
RESULTS
Socio-demographic Characteristics of Participants
Table 20 describes the socio-demographic characteristics of 1440 consecutively sampled adult HIV patients on ART in HTCs in Fako Division. The mean age of the participants was 45.6 (SD = 7.3) years. Of 1440 participants sampled, 1023 (71.0%) were females, 753 (52.4%) were either married or cohabiting and 156 (10.9%) had no formal education. One-fifth of the participants were unemployed. Majority (89.2%) of the participants were Christians. There was a bi-polarization of WHO HIV staging at initiation with 36.6% (stage I) and 26.9% (stage IV). Of the 1440 participants sampled, only 51 (4.7%) were on ART with a protease inhibitor. The mean duration of these HIV patients on ART was 7.3 (SD=2.3). Forty percent of the participants were initiated on ART during the universal test and treat era. One third of the participants had a family history of hypertension.
Table 2: Socio-demographic Characteristics of Participants
|
Variable |
Frequency |
Percentage (%) |
|
Age (Years) |
Mean (SD) |
45.6 (7.3) |
|
21 – 30 |
87 |
6.0 |
|
31 – 50 |
903 |
62.7 |
|
51+ |
450 |
31.3 |
|
Gender |
|
|
|
Male |
417 |
29.0 |
|
Female |
1023 |
71.0 |
|
Marital status |
|
|
|
Single |
414 |
28.8 |
|
Married/Cohabiting |
753 |
52.4 |
|
Divorced/Separated |
219 |
15.2 |
|
Widow(er) |
51 |
3.6 |
|
Level of education |
|
|
|
No formal education |
156 |
10.9 |
|
Primary |
567 |
39.5 |
|
Secondary |
540 |
37.7 |
|
Tertiary |
171 |
11.9 |
|
Employment status |
|
|
|
Employed |
1140 |
79.2 |
|
Unemployed |
300 |
20.8 |
|
Religion |
|
|
|
Christian |
1284 |
89.2 |
|
Muslim |
33 |
2.3 |
|
Others |
123 |
8.5 |
|
WHO clinical stage |
|
|
|
I |
527 |
36.6 |
|
II |
175 |
12.1 |
|
III |
351 |
24.4 |
|
IV |
387 |
26.9 |
|
ART type |
|
|
|
ART with PI |
51 |
4.7 |
|
ART with no PI |
1032 |
95.3 |
|
Duration on ART |
Mean (SD) |
7.3 (2.3) |
|
1 – 5 |
570 |
40.0 |
|
6 – 10 |
516 |
36.2 |
|
11+ |
339 |
23.8 |
|
UTT period |
|
|
|
No |
855 |
60.0 |
|
Yes |
570 |
40.0 |
|
Family history of hypertension |
|
|
|
No |
687 |
64.1 |
|
Yes |
384 |
35.9 |
ART=Antiretroviral therapy, UTT=Universal test and treat, PI=Protease inhibitor, SD=Standard deviation
Prevalence of NCDs among adults HIV patients on ARTs in Fako Division
The overall prevalence of having at least one NCD was 26.9% (95% CI: 24.6 – 29.2). The most commonly observed NCD was hypertension at 18.1% (95% CI: 16.2 – 20.2), followed by osteoporosis at 8.7% (95% CI: 7.3 – 10.5), diabetes mellitus at 4.8% (95% CI: 3.6 – 6.0), renal impairment at 1.9% (95% CI: 1.3 – 2.7), cardiomyopathy at 1.1% (95% CI: 0.6 – 1.7) and asthma at 0.4% (95% CI: 0.1 – 0.9) as shown on figure 2. Prevalence of multi-morbidity was 7.7% (95% CI: 6.4 – 9.2) (Table 3).
Table 3: Prevalence of NCDs among adults HIV patients on ARTs
|
Type of NCD |
Prevalence, n (%) |
95%CI |
|
Overall (1 NCD or more) |
387 (26.9) |
24.6 – 29.2 |
|
Hypertension |
261 (18.1) |
16.2 – 20.2 |
|
Osteoporosis |
126 (8.7) |
7.3 – 10.5 |
|
Diabetes mellitus |
69 (4.8) |
3.6 – 6.0 |
|
Renal impairment |
27 (1.9) |
1.3 – 2.7 |
|
Asthma |
06 (0.4) |
0.1 – 0.9 |
|
Cardiomyopathy |
15 (1.1) |
0.6 – 1.7 |
|
Multi-morbidity (2 or more NCDs) |
111 (7.7) |
6.4 – 9.2 |
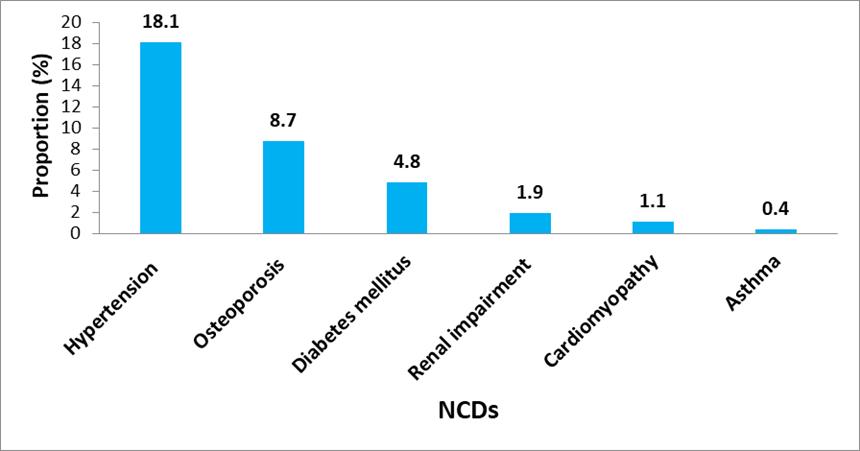
Figure 2: Prevalence of NCDs among adults HIV patients on ARTs
Of the 387 participants with NCDs, 276 (19.2%) had a single NCD, 105 (7.3%) had 2 NCDs and 06 (0.4%) had 3 NCDs (Figure 3).
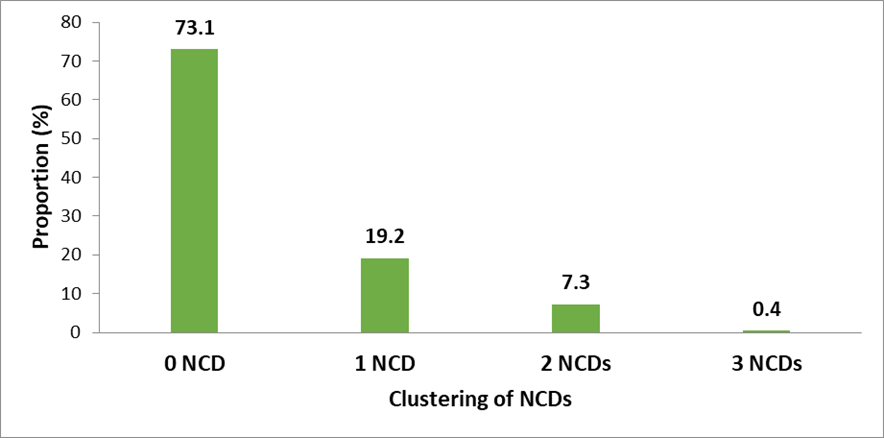
Figure 3: Clustering of NCDs among adult HIV patients on ART
The most common NCD co-morbidities among PLHIV were hypertension-DM (48; 3.3%), hypertension-osteoporosis (39; 2.7%) and hypertension-kidney impairment (15; 1.0%). Interestingly all 69 cases of DM were co-morbid. Three (0.2%) participants had a combination of hypertension-DM-CKD (Figure 4). Asthma seemed isolated as only one case had a co-morbidity with osteoporosis.
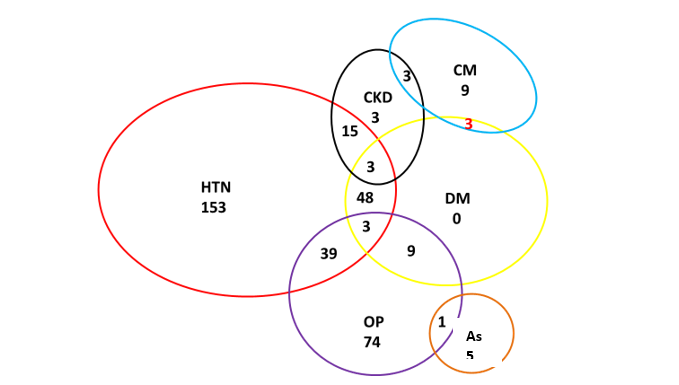
*HTN=Hypertension, DM=Type 2 diabetes mellitus, OP=Osteoporosis, As=Asthma, CKD=Chronic Kidney Disease, CM= Cardiomyopathy
Figure 4: Venn-Euler Diagram showing NCDs co-morbidities in PLHIV
Determinants of NCDs among adults HIV patients on ARTs in Fako Division
After adjusting for confounders in the multivariate analysis, the characteristics that demonstrated a significant and strong relationship with the risk of NCD comorbidity among HIV patients on ART were age (51 years and above) (AOR 3.90; 95% CI 1.50-10.11), tertiary education (AOR 0.25; 95% CI 0.09-0.45), family history of hypertension/diabetes (AOR 5.40; 95% CI 3.69-7.54), psychological stress (AOR 2.33; 95% CI 31.56-3.44), harmful alcohol consumption (AOR 2.61; 95% CI 1.03-4.03), smoking (AOR 3.51; 95% CI 1.96-6.11), baseline WHO clinical stage IV (AOR 4.10; 95 % CI 2.99-5.96) and duration of HIV patient on ART for more than 5 years (AOR 2.03; 95% CI 1.39-2.99) as shown in table 4.
Table 4: Determinants of NCDs among adults HIV patients on ARTs
|
Variables |
NCD co-morbidity |
|
|
Age range (years) |
UOR (95%CI) |
AOR (95%CI) |
|
21 – 30 |
1 |
1 |
|
31 – 50 |
2.36 (1.19-4.98)* |
1.87 (0.78-4.36) |
|
51+ |
5.77 (2.82-11.81)* |
3.90 (1.50-10.11)* |
|
Educational level |
|
|
|
No formal education |
1 |
1 |
|
Primary |
0.64 (0.45-0.89)* |
0.74 (0.41-1.32) |
|
Secondary |
0.36 (0.23-0.55)* |
0.38 (0.23-0.68)* |
|
Tertiary |
0.29 (0.17-0.46)* |
0.25 (0.09-0.45)* |
|
Family history of hypertension/Diabetes |
|
|
|
No |
1 |
1 |
|
Yes |
5.51 (3.16-7.32)* |
5.40 (3.69-7.54)* |
|
Psychological stress |
|
|
|
No |
1 |
1 |
|
Yes |
2.34 (1.55-3.65)* |
2.33 (1.56-3.44)* |
|
Overweight/Obesity |
|
|
|
No |
1 |
1 |
|
Yes |
1.03 (0.56-2.36) |
/ |
|
Harmful alcohol consumption |
|
|
|
No |
1 |
1 |
|
Yes |
2.60 (1.02-4.69)* |
2.61 (1.03-4.03)* |
|
Smoking |
|
|
|
No |
1 |
1 |
|
Yes |
1.93 (1.28-2.99)* |
3.51 (1.96-6.11)* |
|
Physical inactivity |
|
|
|
No |
1 |
1 |
|
Yes |
1.51 (1.12-2.11)* |
1.12 (0.89-1.89) |
|
WHO HIV stage |
|
|
|
I |
1 |
1 |
|
II |
2.64 (1.74-4.02)* |
2.51 (1.62-4.32)* |
|
III |
2.36 (1.66-3.35)* |
2.35 (1.65-3.69)* |
|
IV |
4.07 (2.93-5.63)* |
4.10 (2.99-5.96)* |
|
ART type |
|
|
|
ART with PIs |
1 |
1 |
|
ART without PIs |
1.36 (0.76-2.7) |
/ |
|
Duration on ART |
|
|
|
0 – 5 |
1 |
1 |
|
6 – 10 |
1.51 (1.14-1.98)* |
2.03 (1.39-2.99)* |
|
11+ |
1.35 (0.96-1.79) |
0.71 (0.45-1.23) |
UOR: Unadjusted Odd Ratio, AOR: Adjusted Odd Ratio; CI: Confidence Interval; * = significant variables at p< 0.05, PIs: Protease inhibitors
DISCUSSION
Overall, 26.9% of the participants had a non-communicable disease. These findings are in line with those of other studies carried out in Africa that show non-communicable diseases among people living with HIV range from about 15.0% to 58.0% [12, 13]. However, these studies have looked at different NCDs with some NCDs such as: hypertension, diabetes mellitus, renal impairment, and asthma in common. A lot still needs to be done to give a clearer picture of the NCDs co-morbidity among PLHIV. This is a high prevalence as it implies one in every four adult HIV patients has a non-communicable disease, yet is still an underestimate, as patient files along consultations were used and the actual prevalence is expected to be even higher. Moreover only about half (57.2%) of the HTCs monitor patients for NCDs in Fako Division. Home dispensation of ARVs is common in HTCs in Fako Division, does not offer the opportunity to screen or diagnose NCDs in these patients.
Hypertension was the commonest NCDs identified at 18.1%. This finding is similar to the 15.8% reported in a study among people living with HIV in Uganda [14]. A study carried out in rural Uganda among 65,000 people found a hypertension prevalence of 11.0% in people living with HIV and 14% in people without HIV. This is lower than our study findings and does not suggest differences between people with and without HIV [15].
Osteoporosis was second highest at 8.7% which is comparable to findings from a study carried out in Uganda at the Infectious Disease Institute among a cohort of people living with HIV receiving care at an outpatient HIV clinic which reported prevalence of osteoporosis of about 8 to 9% [16]. This is a high percentage and osteoporosis diagnosis and treatment should be given more priority among HIV patients. Osteoporosis is known to occur in older HIV patients and menopausal women a group that is increasing in PLHIV [16].
Diabetes mellitus was found at 4.8% which is similar to findings from studies among PLHIV in Africa which are mostly below 5.0% according to a systematic review by Haregu et al., (2012) [17]. Our prevalence of diabetes was similar to that in the general community estimated to range from 1 to 4% [18]. However, other studies have reported type II diabetes as being more common among PLHIV. A study in people living with HIV in Nigeria 2016, reported prevalence of diabetes increasing from 2.3% to 5.3% in the first year of being started on ART [19]. The relationship between HIV/ART and diabetes is the subject of ongoing controversy. Observations of both increased risk and no difference have been noted in high income countries. Elevated risks are linked to, among other factors, duration of and exposure to certain ARV drugs like stavudine, efavirenz and protease inhibitors [20]. A systemic review (2013) of the association between HIV/ART and development of T2DM in SSA subjects implied protective effects as judged by reduced HbA1c levels [21]. Notwithstanding, our finding of a 4.8 % T2DM prevalence maybe due to the high prevalence of behavioural and physiological risk factors of DM and the prolonged use of ART. In the general population hypertension and T2DM have similar risk factors: tobacco use, unhealthy diets, physical inactivity and harmful use of alcohol. These have all been documented to be highly prevalent in our study population. ARV drug toxicities including lipodystrophy and dyslipidemias, chronic systemic inflammation, endothelial dysfunction, coagulation disorders and the virus itself are additional risk factors peculiar to HIV disease [22].
Renal impairment was found at 1.9%, which is a low prevalence. However, this finding is similar to findings from a study based on self-reports, carried out in Nigeria [19]. However, this is a much lower prevalence than that from studies that actively screen for renal impairment. A study carried out in Burundi among PLHIV reported 30.0% as being stage I, 13.5% as stage II and 2% as being stage III [23]. Renal impairment is expected to be common, though it is rarely severe, among HIV-infected adults with clinically non-advanced HIV disease [24]. For most individuals it goes undiagnosed until it is severe. Multi morbidity was found at 7.7% where by individuals had more than one non-communicable disease in addition to living with HIV. This finding is quite similar to that reported in Zimbabwe [25] where co-morbidity was found to be 4.5% (95% CI 3.4–8.0) considering hypertension, asthma, type 2 diabetes mellitus, cancer, and congestive cardiac impairment among PLHIV. However, since patients were not actively screened in this study the actual multi-morbidity prevalence is expected to be even higher than what is reported, and interventions targeting this group of people should be considered as challenges such as a high pill burden or higher costs of treatment are more common in this group.
Asthma and cardiomyopathy cases identified were very low at 0.4% and 1.1%. This maybe a result of not actively screening for these conditions in HTCs in Fako Division; and is indicative of the fact that most patients do not know they have certain conditions until they are severe. In addition, this study was carried out at the free outpatient HTCs and patients aware of such conditions are more likely to be in a more intensive care setting such as the inpatient clinic or private patients’ clinic. Though not within the scope of the study to ascertain each included diagnosis, review of some patient case notes indicated that the diagnosis of asthma in some patients was based on adult-onset wheezing, effort dyspnoea and persistent coughing. HIV trained nurses provided the bulk of on-going care including initial patient assessments. Pulmonary function testing was not available either. Consequently, argument can be made to refer to this class of condition broadly as chronic obstructive pulmonary diseases (COPD) [26], which includes chronic bronchitis, fixed airway obstruction, bronchial hyper-responsiveness as well as asthma. COPD has recently come to the fore as the number of persons ageing with HIV continues to grow. Known risk factors for OLD include HIV infection, pulmonary TB, tobacco smoking and indoor pollution from biomass fuel exposure [27]. The latter disproportionately affects women in LMICs, especially SSA. Asthma prevalence among HIV-infected persons is reported to be increased with possibility of a different risk factor profile from the uninfected population [28].
The overall high prevalence of NCDs among HIV patients on ART in Fako could just be the tip of an iceberg as many HTCs were not actively involved in the routine screening for NCDs tracer conditions – hypertension and diabetes. Close to half (42.9%) of the HTCs in Fako Division did not screen routinely for hypertension.
Older people were found to be more likely to have a non-communicable disease (OR = 3.90, 95%CI: 1.50-10.11), which is expected as older people are known to have a much higher risk of non-communicable diseases compared to younger people [25]. The HIV epidemic in SAA is maturing with ever growing numbers of adults ageing with HIV, this is confirmed by the observation from this study of a relatively high proportion (31.3%) being aged 51 years or more. In the general population, NCD risks are known to be considerably increased in this age (50+ years old) group. Indeed, our findings point to age as being an important risk factor for both comorbidity and multi-morbidity in the HIV infected population. Also, people who initiated ART at a later stage of the infection were more likely to have a non-communicable disease as well (AOR = 4.10). This association has been identified by other studies as well according to a systematic review by Haregu T et al in 2012 [17]. Overall NCDs are prevalent among PLHIV, and strategies to encourage early diagnosis and treatment should be intensified.
CONCLUSION
Non-communicable diseases are a growing concern not only due to their health implications, but also their impact on socio-economic status. This study found that 1 in every 4 people living with HIV, has an NCD. Hypertension, osteoporosis and diabetes were the most common NCDs among adult HIV patients on ART in Fako Division. Socio-demographic, behavioural, genetic and clinical factors significantly influenced the occurrence of NCDs among adult HIV patients on ART.
RECOMMENDATION
The Ministry of Public Health should encourage the routine and early screening and diagnosis of NCDs in HTCs. The older patients and those initiated at a later stage of the disease should be given priority. Given the already existing high workload burden on health providers, deployment of community health workers to perform routine screening for non-communicable diseases among people living with HIV should be explored.
STRENGTHS AND LIMITATIONS
The study was conducted in 5 HTCs (a combination of government, private, mission, rural, urban, primary and secondary health facilities), therefore the study findings may be generalizable to people receiving HIV care in Fako Division and other similar settings. A sample size of 1440 was attained, this is representative of the population. This study was carried out at the out -patient clinic where patients received free antiretroviral therapy, thus inpatients and private patients were not included in the study. However, it is expected that most inpatients may have a higher prevalence of co-morbidities. More so patients with known co-morbidities may have been referred to other specialized care facilities. This study also had the limitation of using patient files to determine if an individual has an NCD or not. As much as this gives an estimate of the prevalence of NCDs, the actual NCD burden is higher than what this study estimates. Thus the results need to be interpreted with that in consideration. In similar fashion, it is arguable that those with asymptomatic diseases may have been missed due to the lack of a policy of active NCDs screening in HTCs in Cameroon. Both situations would underestimate comorbidity prevalence. Disease misclassification is a possibility considering that the bulk of diagnoses and on-going care were nurse-driven. Our study did not seek to validate clinical diagnoses. We merely enumerated the diagnosed NCD cases. Non-infectious chronic lung diseases, for example, may have been misdiagnosed and treated as pulmonary TB. We used routine data which is not usually collected with the rigour required for research purposes. Though the data were sufficiently complete allowing us to answer our research question, more detailed data would have enabled additional analyses, case ascertainment and more robust conclusions. Lastly, inclusion of an HIV uninfected age- and gender-matched comparison group would have afforded insights into the putative roles of HIV and ART as drivers of NCDs.
PERSPECTIVES
Further studies should explore and describe the burden of NCDs on adults HV patients on ART in Cameroon.
ABBREVIATIONS
AIDS: Acquired Immuno-deficiency Syndrome; ART: Antiretroviral therapy; COVID-19: Coronavirus Disease 2019; CVDs: cardiovascular diseases; DBP: Diastolic Blood Pressure; eGFR: estimated Glomerular Filtration Rate; HTCs: HIV Treatment Centers; HIV: Human immunodeficiency virus; LMIC: Low- and middle income countries; NCD: Non-communicable disease; PLWH: People living with HIV; PPS: Probability Proportionate to size; SBP: Systolic Blood Pressure; SWR: South West Region; SSA: Sub-Saharan Africa; UTT; Universal Test and Treat; WHO: World Health Organization
FUNDING
This study was self- funded.
AVAILABILITY OF DATA AND MATERIALS
The data that support the findings of this study are available from the corresponding author upon reasonable request.
AUTHORS’ CONTRIBUTIONS
TAY, ALN, NFP and EOG designed the study. TAY, NEN, NJ and KEN did the fieldwork. TAY and TN did the analysis. TAY wrote the first draft, and all authors approved the final version.
COMPETING INTERESTS
The authors declare that they have no competing interests.
CONSENT FOR PUBLICATION
This manuscript does not contain any individual’s data. Consent for publication was obtained from the PhD thesis supervisors.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved for exemption by the Faculty of Health Sciences of the Institutional Review Board at University of Buea.