Journal of Dermatology and Venereology
OPEN ACCESS | Volume 4 - Issue 1 - 2026
ISSN No: 3065-677X | Journal DOI: 10.61148/3065-677X/JDV
Running head: Hair Follicle Stem Cell Disruption in Scarring Alopecia
Madeha Alkelani1*, Henry A. Adeola1, Richard P. Halley-Stott2
1Hair and Skin Research Laboratory, Division of Dermatology, Department of Medicine, Faculty of Health Sciences and Groote Schuur Hospital, University of Cape Town, Cape Town, South Africa.
2Institute of Infectious Disease and Neuroscience, Groote Schuur Hospital, Faculty of Health Sciences, University of Cape Town, South Africa.
*Corresponding author: Madeha Alkelani, Hair and Skin Research Laboratory, Division of Dermatology, Department of Medicine, Faculty of Health Sciences and Groote Schuur Hospital, University of Cape Town, Anzio Road, Cape Town 7925, South Africa.
Received: April 10, 2026 | Accepted: April 18, 2026 | Published: April 22, 2026
Citation: Alkelani M, Henry A. Adeola, Richard P. Halley-Stott., (2026) “Exploring Hair Follicle Stem Cell Involvement in the Development of Primary Scarring Alopecia” Journal of Dermatology and Venereology, 4(1); DOI: 10.61148/3065-677X/JDV/062.
Copyright: © 2026 Madeha Alkelani. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Background: This study investigates the role of hair follicle stem cells in the pathogenesis of primary cicatricial alopecia (PCA), a group of rare disorders that cause irreversible hair loss due to the destruction of hair follicles. The research focuses on identifying the underlying molecular and cellular mechanisms that contribute to scarring and permanent alopecia.
Materials and Methods: We conducted the review using databases such as PubMed, Scopus, ScienceDirect, and Medline, employing search terms including “alopecia,” “stem cells and alopecia,” and “hair follicle stem cells markers.” Relevant studies were selected based on predefined inclusion criteria, and data were extracted from both primary research articles and references. Special attention was given to studies involving immune privilege collapse, stem cell depletion, and inflammation.
Results: Our review identified inflammation in the hair follicle's bulge area as a key factor in the destruction of stem cells, leading to scarring alopecia. Immune privilege in this region, which typically protects hair follicle stem cells, collapses due to autoimmune responses, particularly in disorders like lichen planopilaris (LPP) and cutaneous lupus erythematosus. Studies also reveal the loss of key markers like Peroxisome Proliferator-Activated Receptor γ (PPARγ) in the bulge area, further implicating immune dysregulation in PCA development. Moreover, treatments targeting PPARγ pathways, such as pioglitazone, have shown a reduction in inflammation, indicating potential therapeutic interventions.
Conclusion: The study also explored the molecular changes in the hair follicle stem cells, particularly in response to inflammatory cytokines. Techniques such as immunohistochemistry and fluorescence-activated cell sorting were employed to isolate and analyze stem cells, identifying key markers involved in hair follicle regeneration and scarring processes. Finally, emerging treatments like N-Acetyl-GED-0507-34-Levo (NAGED), which targets PPARγ, demonstrated promise in reversing epithelial-mesenchymal transition EMT and protecting hair follicle stem cells.
The findings suggest that the inflammatory destruction of hair follicle stem cells, coupled with the loss of immune privilege, is a central mechanism in PCA. Further research is required to fully understand the molecular pathways involved and to develop targeted therapies to prevent or reverse scarring alopecia.
Stem cells, Alopecia, Hair follicle, Scarring alopecia, Bulge area, Molecular signature
In recent years, the characterization and identification of adult stem cells has become a topic of great biological and biomedical research interest1. Stem cells, characterized as slow-cycling progenitor cells with dual ability to self-renew and differentiate into the lineage-specific, terminally differentiated functional cells, play a pivotal role in the regeneration of various tissues 2,3. Adult stem cells can be found in virtually all tissues, including blood, muscles, the nervous system, liver, epidermis 3; and ectodermal derivatives such as teeth, feathers, and hair follicles. Hair follicle stem cells, reside in a niche, located in the bulge region of the hair follicle. The bulge is the area located between the opening of the sebaceous gland and the attachment site of the arrector pili muscle 4. Bulge stem cells are multipotent and have high proliferative potential 5. similar to other adult stem cells, bulge stem cells are characterized by their slow-cycling nature, high clonality, and the capacity to generate various lineage specific cells 6, contributing, for instance, to the generation of skin epithelial cells, including sebocytes, hair and keratinocytes.
The consequences of hair loss extend beyond physical implications, impacting self-esteem, self-confidence, and leading to increased levels of anxiety and depression. Patients with scarring alopecias, experience a considerable reduction in the quality of life and high levels of distress 7,8. Primary scarring alopecia affects both women and men, most commonly adults of a wide age range and ethnicities and it can be permanent or temporary; however, it is relatively rare in children. It is neither life threatening nor painful, though there can be irritation of the skin, as well as physical problems resulting from the loss of eyelashes and eyebrows 9-11. This form of irreversible hair loss is characterized by the disappearance of visible follicular ostia within an area of alopecia and histologically by the destruction of hair follicles and their replacement with fibrotic tissue 12,13. Scarring alopecia can be classified into two categories, primary or secondary. Primary cicatricial alopecia (PCA) is very challenging in terms of classification and diagnosis, both for clinicians and pathologists. In secondary cicatricial alopecia, destruction of the hair follicle is incidental to a non-follicle-directed process or external injury, such as severe infections, burns, radiation, tumors, or traction 14. This review is confined to PCA, in which the hair follicle is the target of the destructive inflammatory process.
Method
Database and resources including PubMed, Scopus, ScienceDirect, Medline, and google scholar were systematically searched from the earliest available online indexing up to 2023. The search criteria included keywords such as “alopecia”, “stem cells and alopecia”,
“the role of stem cells in alopecia”, “hair follicle stem cells markers”, and “pathogenesis of alopecia”. Additionally, references
cited in identified articles were read to obtain extra information. The initial screening involved evaluating the title and abstract of all retrieved studies, followed by a thorough review of the full texts of potentially relevant studies. The inclusion and classification of studies were determined based on predefined criteria, and pertinent data were subsequently extracted from the selected studies.
Hair follicle stem cells
Hair follicle stem cells constitute the primary source of multipotent stem cells in the skin 1. These stem cells, resident in hair follicles and sebaceous glands, play a crucial role in repopulating the epidermis after injury. During the production of a hair, the follicle undergoes dynamic changes from an actively growing phase (anagen) to a remodeling phase (catagen) and eventually entering a quiescent or rest phase (telogen), before repeating the cycle. Two key elements controlling hair follicle cycling are the follicular epithelial stem cells and the specialized mesenchymal cells that constitute the follicular (or dermal) papilla 15. Hair follicle stem cells control the growth of hair.
Historically, hair follicle stem cells were assumed to be located exclusively in the secondary germ Figure 1, which resides at the base of the telogen hair follicle. The secondary germ was moved downward to the hair bulb during the anagen phase, provided new cells for hair production. At the end of anagen, the secondary germ moves upward with the dermal papilla during catagen to come to rest at the end of telogen club hair 16. Subsequently, it has been revealed that the secondary germ is a transient structure that forms at the end of catagen from cells in the lower bulge 17. Lineage analysis has since confirmed that hair follicle stem cells are permanently located in the bulge, capable of giving rise to all epithelial layers of the skin, sebaceous glands, and serving as the exclusive source material for hair follicles The concept that hair follicle stem cells are permanently located in the bulge has now been confirmed using lineage analysis, which demonstrated that the bulge cells are able to give rise to all epithelial layers of the skin and sebaceous glands, and exclusively provide the source material for hair follicles 5,18,19. In line with this, ablation of bulge cells results in destruction of the hair follicle 17. These findings support the concept that loss of hair follicle stem cells in the bulge region leads to irreversible or permanent scarring type of alopecia. Microscopically, the bulge region is identified as a prominent protuberance below the sebaceous gland in vertical section of murine and human fetal specimens stained with hematoxylin and eosin. Unlike the bulge of murine follicles, the human adult anagen bulge is slender, making it difficult to see in human skin. However, characteristic protrusions (the follicular trochanter) can provide a useful histological demarcation of the human bulge 20.
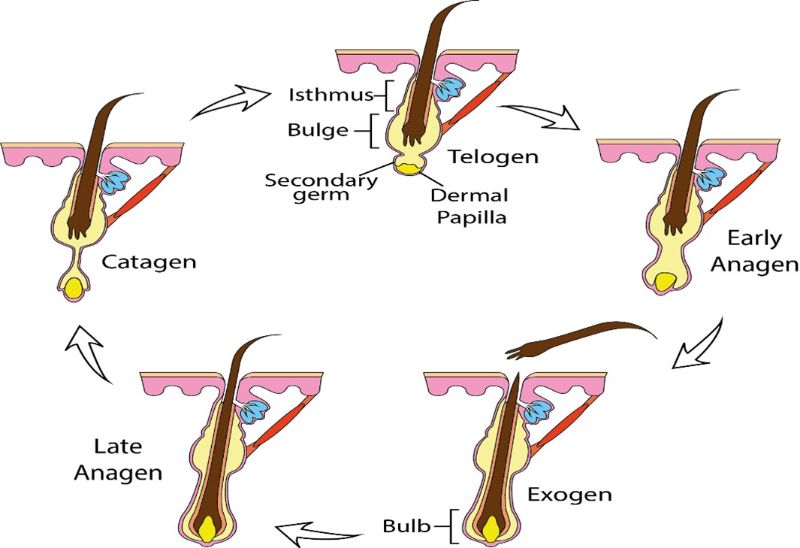
Figure 1: Hair follicle growth cycle and anatomy.
Hair follicle stem cells markers
To identify and isolate stem cells, the identification of specific stem cells markers is essential. To date, the most reliable markers for bulge stem cells in murine hair is cytokeratin CK1521,22. Antibodies against CK15 preferentially stain the outer root sheath around the insertion point of the arrector pili muscle 23. Initially confirmed in hair follicle bulge cells in human scalp, CK15 has also been shown to be expressed in adult mouse bulge cells. Human bulge cells have been found to selectively express CK15 in all stages of the hair cycle 24,25. CD34, a cell surface protein uniquely expressed in mice 26, serves as a marker for mouse bulge cells. CD34 is reported to be uniquely expressed in mouse hair follicle bulge keratinocytes, facilitating the isolation of living keratinocytes with stem cell SC characteristics from mouse hair follicles. However, CD34 is not significantly enriched in human bulge cells27. Hence, CD34 represents the best marker for mouse hair follicle bulge cells, providing a valuable tool for studying bulge cell biology 5.
Nestin, an intermediate filament typically associated with neural stem cells, has been suggested to be expressed in bulge cells 28,29. Nestin is expressed in bulge-area stem cells of the hair follicle.
CD200 is a cell-surface marker involved in autoimmunity 30. It is a biomarker for human follicular stem cells that reside in the bulge 4,31. It has been reported that CD200 is more specific in detecting human bulge stem cells than CK15 32.
Microarray analysis in a study investigating cell surface marker for bulge stem cells revealed a panel of cell surface markers for human bulge cells. CD200 and CD59 are up-regulated in bulge compared to other defined hair follicle regions, while CD24, CD34, CD7 and CD146 were down-regulated. In addition, several cell surface proteins, including follistatin, PHLDA1 and frizzled homolog 1 (FZD1), were up-regulated in human bulge cells 33.
Scarring alopecia’s
Primary scarring alopecias (PSAs) are commonly attributed to irreversible damage to the hair epithelial stem cells that reside in the hair follicle 9. However, there are limited studies regarding the pathogenesis of scarring alopecia in humans 34. A multitude of factors have been implicated in triggering this group of rare disorders, which lead to the destruction of the hair follicle, replacement with scar tissue, and result in permanent hair loss. Unfortunately, PCAs are frequently difficult conditions to diagnose and treat 9. Furthermore, the pathogeneses of each of the diseases within this group remains poorly defined in many instances 35,36. Hypothesized theories are based on autoimmune origins, sebaceous glands gene expression changes, loss of immune privilege, and destruction of the bulge stem cells region 37. Considering that scalp biopsy is often non/poorly-diagnostic, a comprehensive clinical examination and accurate history taking are crucial for diagnosing primary cicatricial alopecia 38. Clues for accurate diagnosis may be obtained from factors such as age at inception, history of other associated skin diseases such as lupus, lichen planus, or other autoimmune disease, and earlier hair-care practices, along with signs and symptoms such as itching or burning of the scalp and purulent discharge 39.
The etiologies and subsequent development of cicatricial alopecias are not fully understood but are widely believed to be autoimmune disorders that arise from a combination of genetic and environmental influences 40. An inflammatory infiltrate is typically observed in the immediate vicinity of affected hair follicle HFs, and histological examination reveals a composition suggestive of the specific type of PCA present (Table 1). The current classification of PCAs relies on the predominant inflammatory infiltrate, categorizing disorders as lymphocytic, neutrophilic, mixed, or nonspecific. Further sub-classification is determined by the expressed clinical features 41. Supplemental diagnostic evidence can be acquired from immunofluorescence, especially if cutaneous lupus erythematosus or bullous dermatoses are suspected.
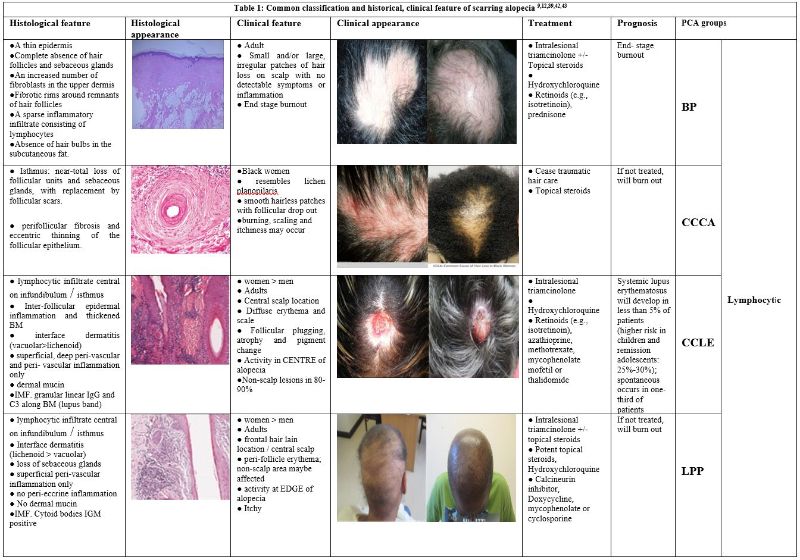
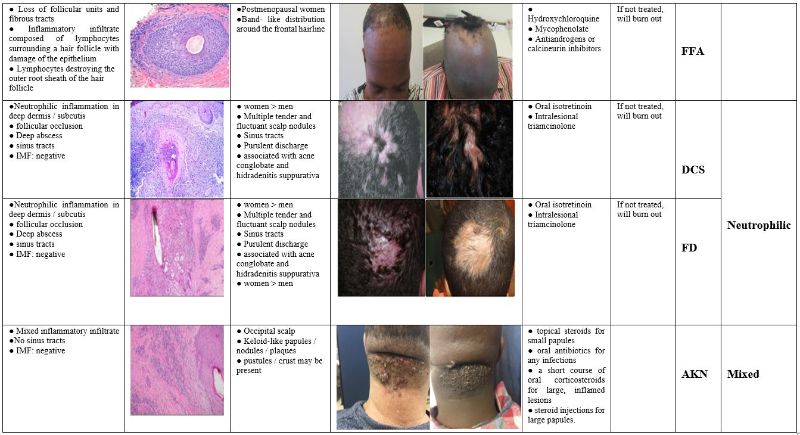
Roles of stem cells in alopecia
There is indirect evidence in humans and direct evidence in mice that compromising the integrity of the bulge and/or sebaceous gland is important in the development of alopecia. A key difference between the scarring and reversible alopecias lies in the location of the perifollicular inflammatory infiltrate relative to key structures of the hair follicle, specifically the hair bulge and hair bulb. In scarring alopecia, the infiltrate surrounds the bulge region and distal (permanent) follicle, whereas in reversible alopecias, the infiltrate is typically confined to the transient structures of the proximal follicle.
The loss of hair follicle stem cells appears to be a key element in the pathogenesis of primary scarring alopecia. this is supported by the observation that the inflammatory cell infiltrate in most PCAs predominantly surrounds the bulge region and distal follicle, sparing the proximal follicle. Moreover, K15, a marker of bulge cells in humans has been shown to be reduced in the bulge region in lichen planopilaris LPP and CCLE (chronic cutaneous lupus erythematosus)44. Fundamentally, models with dense peri-follicular inflammatory infiltrates suggest that the inflammatory process either destroys the hair follicle stem cells pool or induces these cells toward differentiation as part of a HF repair response. Regardless, even if the inflammatory process does not directly target the hair follicle stem cells, a significant insult can overwhelm the regenerative potential of the stem cells, leading to "stem cell exhaustion" and permanent hair loss.
There have been limited studies on the pathogenesis of scarring alopecias in human, especially regarding the role of stem cells in the scarring process. Patients with scarring alopecia typically seek medical advice at a late disease stage, and this may limit the histopathological study of their lesions. However, using a mouse model of discoid lupus erythematosus DLE, decreased proliferation (by Ki-67 expression as a proliferation marker) in bulb matrix cells and increased apoptosis have been observed45.
Furthermore, immune-mediated destruction of bulge stem cells plays a crucial role in the pathogenesis of alopecia in a specific mouse strain (the alopecia and excoriation mouse: AE strain), and this might be an interesting model for scarring, especially for LPP. AE mice exhibit a progressive alopecia phenotype similar to that seen in primary scarring alopecia’s, showing hair loss on the head from postnatal day 22 and complete hair loss by the age of 6 months. Consistently, immune privilege collapse, stem cell destruction, and the alopecia phenotype of AE mice were all rescued after treatment with the immunosuppressive agent ciclosporin. This suggests that immune-mediated destruction of bulge stem cells may play a central role in the pathogenesis of this alopecia in a specific mouse strain 46.
LPP is one of the inflammatory scarring alopecia which has been somewhat studied for the role of the bulge region in the pathogenesis of the scarring process. Histologically, in the inflammatory stage, LPP is characterized by the presence of a band-like lymphocytic infiltrate at the bulge region with sparing of the lower portion of the follicle. Consequently, Mobini et al. 47 investigated the possible role of the bulge region in the pathogenesis of this inflammatory scarring alopecia, in a case series of 35 patients with LPP. They demonstrated a marked decrease in proliferating stem cells, highlighted by Ki-67, in the bulge compared to uninvolved follicles or normal control. Additionally, their study showed a significant perifollicular lymphocytic inflammatory infiltrate, predominantly of a cytotoxic type. Furthermore, CK15 immunostaining was shown to be reduced or absent from the bulge area in LPP.
Other types of scarring alopecia, including lupus erythematosus, dissecting cellulitis, folliculitis decalvans, and follicular degeneration syndrome, were examined by immunohistochemistry using CK15, nestin and CD34 as stem cells markers. These studies supported previous findings about stem cell involvement by showing the absence of CK15 and CD34 from 53% and 69% of scarring alopecia samples compared with non-scarring type (present in all cases) 24.
The pathogenesis of scarring alopecia and its relationship to stem cell association is a complex process. Irreversible damage to epithelial stem cells is believed to be primarily due to inflammatory mechanisms, particularly in the context of autoimmune disease such as cutaneous lupus erythematosus, where the hair follicle itself is the key target of auto destructive immunity 9. In addition, other factors may contribute, including the loss of immune protection of stem cells, impaired stem cell self-maintenance, and enhanced autoimmunity caused by proinflammatory cytokines and environmental/genetic pre-dispositions 48.
Hair follicle stem cells isolation
Investigating hair follicle stem cell biology and their applications in clinical settings, such as gene therapy or regenerative medicine, would be significantly facilitated by the isolation of living bulge stem cells 33. Microdissection has been employed for isolating bulge stem cells from the hair follicles to study the morphology of stem cells 49,50, however, this technique is time-consuming, requires skills, and isolation of a uniform cell population is difficult. The identification of bulge stem cells specific markers would qualify a more accurate and high-throughput isolation of those stem cells, greatly aiding further investigation 51.
Although CK15 is a potential marker, it is not adequately restricted to the stem cell lineage in humans for practical use 5,33. GFP driven by the CK15 promoter has proven successful in isolating bulge cells through fluorescence-activated cell sorting (FACS). Similarly, slow cycling bulge cells have been effectively isolated using inducible live labeling with histone H2B-GFP fusion proteins.
Changes to the phenotype and molecular profile of stem cells
Peroxisome Proliferator-Activated Receptor γ (PPAP-γ)
Karnik et al. carried out a study 52, reporting that the loss of PPARγ expression in the stem cells of the bulge area resulted in a LPP-like phenotype with progressive hair loss, sebaceous glands atrophy, scarring, and inflammation in a mouse model. These observations strongly implicate PPARγ defects in the pathogenesis of (some) scarring alopecias. Their study demonstrates that perturbation of lipid metabolism induced by PPARγ deficiency, most likely an acquired condition, results in inflammation-induced destruction of the pilosebaceous gland in scarring alopecia. These effects suggest several crucial roles for PPARγ in the maintenance and normal functioning of the pilosebaceous unit and suggest that loss of this signaling pathway may be responsible for the pathogenesis of scarring alopecia 52. This work has led to the therapeutic trial of the PPARγ agonist pioglitazone, with a 50% reduction in LPP-associated inflammation 53,54. Furthermore, PPARγ signaling down-modulates inflammatory responses 55 and up-regulates expression of K15 in the bulge of normal human scalp HFs ex-vivo 55. Therefore, PPARγ stimulation appears to supper, or even partially reverse, bulge epithelial–mesenchymal transition, in an epithelial hair follicle stem cell- protective (eHFSC) and immunoinhibitory manner.
In line with the above thinking, Pirat et al.56 developed a new PPAR-γ modulator with agonistic activity, N-Acetyl-GED-0507-34-Levo (NAGED)56. The interest in NAGED is based on its potential application to the management of LPP, as it stimulates the expression of the stem cell-associated K1557 and protects hair follicle stem cells from experimentally-induced EMT 58 in human scalp HFs ex vivo. Furthermore, NAGED can moderately reverse the EMT signature in the bulge of lesional LPP hair follicle ex vivo 58.
In a recent investigation, organ-cultured lesional scalp samples from two LPP patients with lymphocytic inflammatory cell infiltrates in/around the isthmus were treated with vehicle or 0.1 mmol/L NAGED59. The number of K15+ HFeSC and K15 protein expression is higher in lesional LPP hair follicle bulges than in vehicle HFs. This early finding revealed that NAGED may prevent and partially reverse K15+ HFeSC depletion in LPP patients59. NAGED treatment decreased the number of CD8+ T cells, the essential pathogenic T cells in LPP, and MHC class II+ cells around/in the epithelial bulge cells, indicating that NAGED lowers the inflammatory infiltration attack on the bulge and may partially restore bulge immunological privilege59.
Immune privilege collapse
Harries MJ and colleagues conducted a study in 2010 60 to investigate if collapse of immune privilege in the hair follicle plays a role in the pathogenesis of primary cicatricial alopecia. The collapse of immune privilege (IP) in the hair follicle bulge is a striking theory that seeks to explain the exposure of eHFSCs to immune-mediated attacks in primary cicatricial alopecia. The study found that immunohistochemical investigation increased the expression of major histocompatibility complex (MHC) classes I and II and o β2-microglobulin in the bulge region of lesioned follicles compared with uninvolved follicles.
An additional study aimed to investigate whether the human HF bulge is a site of relative IP within mature HFs. They reported that MHC class Ia, ß2- macroglobulin, and MHC class II immunoreactivity are downregulated in the human bulge 61. The immunosuppressants, including -melanocyte stimulating hormone, transforming growth factors-ß2, macrophage migration inhibitory factor, and indoleamine-2, 3-dioxygenase 23. Therefore, the collapse of immune privilege induced by interferon-gamma (IFN-γ) likely lies at the heart of LPP/ frontal fibrosing alopecia FFA pathobiology 62.
Another mechanism involved in hair follicle immune privilege is the "no danger" signaling via the type-1 transmembrane glycoprotein CD200, which is prominently expressed in the bulge region61. The interaction of CD200 and its receptor, CD200R significantly diminishes antigen presenting cell (APC) activity and the secretion of proinflammatory cytokines by activated T cells63. CD200-CD200R interaction is thought to promote tolerance and prevent autoimmunity within the HF64.
Environmental factors
Environmental factors, such as “leave-on” cosmetics, have been shown to be associated with FFA65. A study reported a high frequency of the use of facial sunscreen in FFA patients compared with controls, with most FFA patients testing positive to fragrances on patch tests, especially to linalool, hydroperoxide of limonene, and balsam of Peru66. The authors applied the patches to the back of the patients (40) using Finn TM chambers on Scanpor TM tape66. Readings were collected after 48 and 96 hours66. Despite the high positive results for FFA patients, no association between FFA and facial products, hair care products, and hairstyle procedures was proven66. This study estimates that the cause for FFA might not be a specific ingredient of facial products, such as the UV filters, but rather their retention within the hair follicle once applied to the skin probably lead to low sebum production in the affected patients. An increased prevalence of sensitization to fragrance could reflect greater use of cosmetics in the FFA population, as suggested by another study67. This extended retention could trigger an immunological response68. In addition, a recent cross-sectional study of 72 women diagnosed with FFA reported that tobacco exposure could play a role in the development of FFA.
Genetic and family background
The genetic aetiopathogenesis of FFA has been hypothesized but not conclusively proven70,71. FFA has been diagnosed in siblings and relatives, suggesting a genetic basis70,72. Reports of familial cases 73,74 and data from case series suggest that 5-8% of reported FFA cases display a positive family history72,75,76, with links to specific human leukocyte antigen (HLA) alleles (HLA-DRB typing revealed DRB1*04:XX,11:XX, DRB3: positive, DRB4: positive, and DRB5: negative, HLA DRB1FNx0104,13 and DQB1FNx0103:02,06) being identified in some cases77-79. The genetic aetiopathogenesis of FFA has been hypothesized but not conclusively proven70,71. FFA has been diagnosed in siblings and relatives, suggesting a genetic basis70,72. Reports of familial cases 73,74 and data from case series suggest that 5-8% of reported FFA cases display a positive family history72,75,76, with links to specific human leukocyte antigen (HLA) alleles (HLA-DRB typing revealed DRB1*04:XX,11:XX, DRB3: positive, DRB4: positive, and DRB5: negative, HLA DRB1FNx0104,13 and DQB1FNx0103:02,06) being identified in some cases77-79.This clarifies the family association, albeit currently rare, with the late onset of FFA and suggests an autosomal dominant inheritance pattern with incomplete penetrance or a germline predisposition in women80, 81. Furthermore, the fact that FFA develops later in life suggests that environmental factors such as cosmetic products, allergens, chemical exposure, or food may play a role in the development of this disease71. Epigenetic factors and miRNA signaling may also play a role 82. Again, once the role of genetic and epigenetic factors in the shared and distinct elements of epithelial stem cells eSC pathology that underlies FFA versus LPP has been clarified, this invites scrutiny of other human eSC pathology for abnormalities in the expression/activity of the same genes and pathway 54. A UK-based genome-wide association study on FFA is currently underway that may help to clarify the role of genetics in this disorder81,83.
Functional analyses revealed the involvement of immune response pathways and fatty acid metabolism in the disease process. Further investigations highlighted the activation of M1 macrophages and CD8+ T cells, suggesting a significant immune component. Additionally, ferroptosis—a form of regulated cell death driven by iron-dependent lipid peroxidation—was identified as a potential mechanism contributing to the pathophysiology of FFA84. In exploring the role of genetics in alopecia, Malki et al85. conducted an exome sequencing central centrifugal cicatricial alopecia CCCA case-control study85. The study identified one splice site and three heterozygous missense mutations (c.856A→G, c.1744G→A, c.1669C→T, and c.832-2A→G) in PADI3 in CCCA patients (31%). The approximate prevalence of the disease was reported to be 5.6% in women of African ancestry85. PADI3 encodes peptidyl arginine deiminase, type III (PADI3), an enzyme that post-translationally modifies other proteins essential to hair-shaft formation. These mutations were found to result in reduced PADI3 expression, abnormal intracellular localization of the protein, and decreased enzymatic activity-findings that support the role of PADI3 in CCCA pathogenesis85.
Immunofluorescence staining demonstrated decreased PADI3 expression in CCCA scalp biopsies85. The study further directly sequenced PADI3 in 42 patients and observed genetic variants in 9 of them85. Their data showed that the prevalence of PADI3 mutation was higher among CCCA patients as compared with the control cohort of women of African ancestry. Mutations in PADI3, which encodes a protein that is essential to proper hair-shaft formation, were associated with CCCA85.
Neurogenic and hormonal factors
A recent study reported increased scalp sweating in some patients with FFA86. However, the authors were unable to state if both conditions are associated, and they suggested more investigation be conducted on neurogenic inflammation86. Antisweat therapy decreased scalp irritation and inflammation, stabilizing the illness. The study postulated that FFA's inflammatory process may cause or regulate sweating. Increased forehead and frontal hairline perspiration may start and maintain hair follicle irritation86. Botulinum toxin injections alleviated increased perspiration and FFA symptoms in two patients86.
FFA predominantly affects postmenopausal women87. It responds to 5α-reductase inhibitor (5AR), and is frequently associated with androgenetic alopecia87. Hormones have been postulated as possible triggers. Androgens such as dehydroepiandrosterone (DHEA) have been argued to have a major role in the pathogenesis of FFA as it is pointed out that DHEA and DHEA-sulphate are essential for PPAR-γ activity, and their decline with menopause could explain the PPAR-γ deficiency describes in FFA87. In spite of this, a retrospective study associated FFA with androgen deficiency, compared with LPP, which was more frequently associated with androgen excess88. Consequently, the role of hormones is uncertain and still questioned. What is known is that FFA occurs in both pre- and postmenopausal women, is somewhat rare in males, considerably affects androgen-independent hairs such as the eyebrows, is unaffected by hormone replacement therapy, and has an increasing incidence since its first description in the early 1990s89.
The molecular signature of hair follicle stem cells
In lesional LPP/FFA HFs, the bulge undergoes major pathological changes, including reduced protein expression of the key bulge stem cell markers K15 and CD200 24,44. Furthermore, microarray analysis of mRNA extracted from laser-capture microdissected bulge epithelium from lesional human LPP HFs shows a loss in the expression of eHFSC signature genes, and K15+ cells in the bulge of lesional LPP and FFA HFs undergo increased apoptosis in situ 62.
Microarray analysis, using laser capture microdissection for targeted extraction of mRNA from HF bulge cells, revealed increased mesenchymal gene expression signatures in LPP 62,90. Imanishi et al. 58 showed that bulge epithelial stem cells indeed attain a fibroblast-like phenotype in lesional LPP HF histologically, ultrastructurally, and by immunohistology. Moreover, the epithelial marker E-cadherin was greatly reduced, whereas key mesenchymal markers and transcription factors were up-regulated in the bulge compartment 58. Subsequently, they were able to demonstrate that stimulation with an EMT-promoting ‘‘cocktail’’ (comprising transforming growth factor beta 1, interferon gamma, epidermal growth factor, and the E-cadherin antagonist peptide A) was enough to rapidly induce an EMT gene and protein expression signature in the bulge of healthy, organ-cultured scalp HFs, thus implicating 4-key drivers of EMT induction. Moreover, these experimentally induced EMT changes could be prevented by the peroxisome proliferator activated receptor gamma agonist pioglitazone, which is already used in the clinic for managing LPP, and a topically applicable modulator, N-acetyl guanidinoethyl disulfide 58.
Conclusion and perspectives
Patients with scarring alopecia typically seek medical advice at advanced stages, limiting histopathological studies of lesional biopsies and effective treatment options. We have also discussed various techniques for human hair follicle isolation and characterization. More studies on scarring alopecia are imperative to comprehensively understand major pathological changes, including protein expression and gene alterations, throughout the stages of the disease. Ongoing research efforts will contribute to enhanced diagnostic and therapeutic strategies for individuals affected by PCAs.
PCAs represent a group of inflammatory diseases characterized by progressive and permanent destruction of hair follicles followed by replacement with fibrotic tissue. Unfortunately, when patients seek assistance for hair loss, the skin is often already inflamed and/or scarred, diminishing the likelihood of a return to normal hair growth patterns. Healthy HFSCs appear to benefit from relative protection from inflammatory assaults by being in an immunologically “privileged” niche. Evidence suggests that this protection may collapse in prevalent forms of PCAs, such as CCLE and LPP. The restoration and maintenance of eHFSC immune privilege, possibly through normal PPAR-γ receptor-mediated signaling, are crucial to maintaining healthy hair follicles in these conditions.
A better understanding of the pathobiology of the predominate (lymphocytic) PCAs will allow the development of more effective PCA treatments. Animal models, have long been used as a proxy for human disease and also for preclinical therapeutic testing, have provided valuable insights into the role of stem cells in PCA pathogenesis. The pathogenesis of scarring alopecias and the destruction of hair follicle stem cells involve complicated processes. In order to get a clearer understanding of the role that stem cells play in these diseases, more human studies need to be performed. We also discussed the different techniques for human hair follicle isolation and characterizations. More studies on scarring alopecia must be performed to give a clear understanding of major pathological changes such as proteins expression and gene changes during the stages of the disease.
Acknowledgement: HAA thanks the South African Medical Research Council (SAMRC) for a mid-career scientist and Self-initiated research grant; and the NRF for a research development grant for rated researchers. NPK : SARChI Chair (in Dermatology and Toxicology) and the MRC EM Unit (Wound Healing and Keloid Scarring) The funders played no role in the design or interpretation of the paper or the decision to publish.
Conflict of Interest: The Authors declare that they have no conflict of interest.
Authors’ contributions: MA and HA conceptualized and designed the study. MA, HAA and RPH prepared and critically revised the manuscript, tables, and figures. All authors read and approved of the final manuscript.