International Journal of Medical Case Reports and Medical Research
OPEN ACCESS | Volume 5 - Issue 1 - 2026
ISSN No: 2994-6905 | Journal DOI: 10.61148/2994-6905/IJMCRMR
Giacomo Manfredi
Former Head, Allergy and Clinical Immunology Unit.
Department of Clinical Medicine, E.E. Ospedale Gen. Reg. Miulli, Acquaviva delle Fonti (BA).
*Corresponding author: Giacomo Manfredi, Former Head, Allergy and Clinical Immunology Unit.
Department of Clinical Medicine, E.E. Ospedale Gen. Reg. Miulli, Acquaviva delle Fonti (BA).
Received: May 02, 2026 | Accepted: May 14, 2026 | Published: May 20, 2026
Citation: Manfredi G., (2026) “Chronic Urticaria’s Aetiology: Enlightening The Dark Side Of The Moon.” International Journal of Medical Case Reports and Medical Research, 5(1); DOI: 10.61148/2994-6905/IJMCRMR/0179.
Copyright: © 2026 Giacomo Manfredi. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Urticaria is characterized by wheals, erythema, and sometimes localized angioedema. It is defined as chronic (CU) when symptoms persist for more than 6 weeks; however the aetiology is incompletely understood. The aim of this study was to find causes and best treatments of CU.
For this purpose, personal and clinical data from 100 patients with CU were collected. The diagnostic work-up included stool parasite testing and the blood tests: IgG antiviral titres for EBV, CMV, HSV1, VZV and COVID-19; IgE for Anisakis; diamine oxidase (DAO); genetic testing for lactase deficiency; C3, C4; complete blood count; serum electrophoresis; anti-TG, anti-TPO, antinuclear and anti-dsDNA antibodies; P-ANCA, C-ANCA. Blood IgG antiviral Titers/Threshold Ratio (aVGt/TR) was evaluated in patients and 100 healthy controls (50 Male, 50 Female, 15–80 y.o.), divided into two groups, above and below 10 times the threshold value; statistical significance was assessed using the χ² test. Clinical outcomes, evaluated as changes in the UAS7 score before and after therapy, were categorized as: A) Healing; B) Improvement (decrease >70%); C) Unchanged (decrease <70%).
CU showed a M/F ratio = 0.52. Viral reinfections, identified by an aVGt/TR >10 (p < 0.0005), were the most frequent aetiological cause (71%), followed by autoimmunity (16%), anisakiasis (13%), histamine intolerance/DAO deficiency (3%), physical factors (2%), and idiopathic causes (2%). With targeted therapies, the patient-reported outcomes were: A) Healing 74%; B) Improvement 20%; C) Unchanged 6%.
CU aetiological diagnosis was achieved in 95% of cases. The term “spontaneous” chronic urticaria should be reserved for the refractory patients.
Urticaria is characterized by wheals, erythema, and sometimes localized angioedema. It is defined as chronic (CU) when symptoms persist for more than 6 weeks; however the aetiology is incompletely understood. The aim of this study was to find causes and b
Chronic urticaria has so far been classified (EAACI, WAO) on the basis of clinical criteria
-spontaneous and inducible- since attempts to identify its aetiology have often been unsuccessful (9). It is a disabling pathology, significantly impairing quality of life and interpersonal relationships (4,9). Recognized causes include cutaneous mastocytosis, mast cell activation syndrome, and autoimmune vasculitis; however, many syndromes presenting with chronic urticaria remain of unknown aetiology.
Symptoms such as pruritus, wheals, erythema, and angioedema derive from activation of cutaneous epithelial receptors (H1R and LTR), vasodilation, and increased vascular permeability.
They are attributable to the local release of mediators including histamine, leukotrienes, prostaglandins, heparin, chemokines (e.g., TNFα and IFNβ), cathelicidins, and proteases, with activation of kinins (52,54). These mediators are stored in skin mast cells, which in connective tissues are typically equipped with both chymase and tryptase (MCTC), whereas mucosal mast cells contain mainly tryptase (MCT) (54).
Mast cells are effectors of both innate and adaptive immunity and can transition from a resting to an activated state in response to multiple triggers able to prompt degranulation with release of preformed mediators and synthesis of newly produced ones (2,5,6,8,32,55). Triggers include IgE–allergen immune complexes, IgE–autoantigen immune complexes, IgG–bacterial and IgG–viral immune complexes, IgG–autoantigen immune complexes, RNA and DNA viruses, and bacterial peptidoglycans. Mast cells express pattern-recognition receptors, including Toll-like receptors (TLRs) and Pathogen Associated Molecular Patterns (PAMPs), whose engagement can induce activation and degranulation (27).
Skin keratinocytes challenged by viruses and bacteria (27) produce mediators including alarmins such as Thymic Stromal Lymphopoietin (TSLP) and IL-33, and growth factors such as Stem Cell Factor (SCF), which engages the c-kit receptor and results in mast cell proliferation and activation. IL-33 can induce synthesis and release of cytokines such as TNFα and IFNβ, which exert antibacterial and antiviral effects, and it also increases expression of ICAM-1 (Fig. 1). On mucosal mast cells, ICAM-1 can facilitate engagement by rhinoviruses, enhancing cellular invasion (30).
Circulating histamine is also produced by intestinal enterochromaffin cells during digestion (46,47), and in some cases its increase may be due to impaired catabolism by diamine oxidase (DAO), an enzyme produced by the epithelium covering the intestinal villi (7,42,43). This reduction may be primary or secondary and can lead to histamine intolerance. The aim of this study is therefore to identify the causes of chronic urticaria and the most effective treatments for a condition that remains only partially explained.
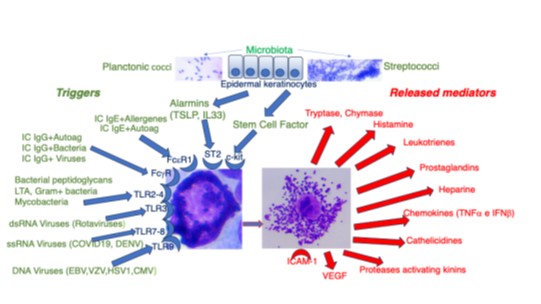
Fig1 Mastocytes degranulation’s triggers in Chronic Urticaria.
Materials and Methods
Subject to informed consent, computerized data were collected from 100 patients with chronic urticaria examined over the last 10 years, recording age, sex, identified aetiology, comorbidities such as gut microbiota subversion due to disaccharide intolerance, administered therapies, and patient‑reported clinical outcome. The diagnostic pathway involved evaluation of IgG antiviral titres (EBV, CMV, HSV1, VZV, and COVID‑19), specific IgE for Anisakis, DAO, genetic testing for lactase deficiency, C3, C4, complete blood count, serum electrophoresis, anti‑TG and anti‑TPO antibodies, antinuclear antibodies, anti‑dsDNA antibodies, P‑ANCA, C‑ANCA, and stool parasite testing. Anti‑viral IgG Titers/Threshold Ratio (aVGt/TR) values exceeding 10 times the threshold were distinguished from those below it, and the data were evaluated in patients and in 100 healthy controls (50 men, 50 women, 15–80 years old); the results were statistically analyzed using the χ² test. Clinical outcomes were assessed through the patient‑reported UAS7 score before and after the administered therapies and were divided into three categories: A) Healing; B) Improvement (decrease >70%); C) Unchanged (decrease <70%).
Results
Chronic urticaria affected women approximately twice as frequently as men (M/F ratio = 0.52) and showed no age preference (11–81 years) (Fig. 2).
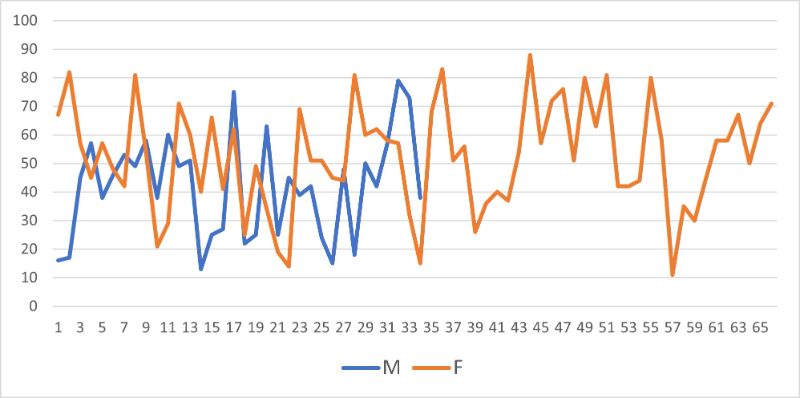
Fig.2 Age and Sex prevalence in 100 patients suffering from Chronic Urticaria
The aetiology included most frequently viral reinfections as responsible or jointly responsible (71%), followed by autoimmune causes (16%), anisakiasis (13%), DAO deficiency/histamine intolerance (3%), physical causes (2%), and spontaneous urticaria (2%) (Fig. 3).
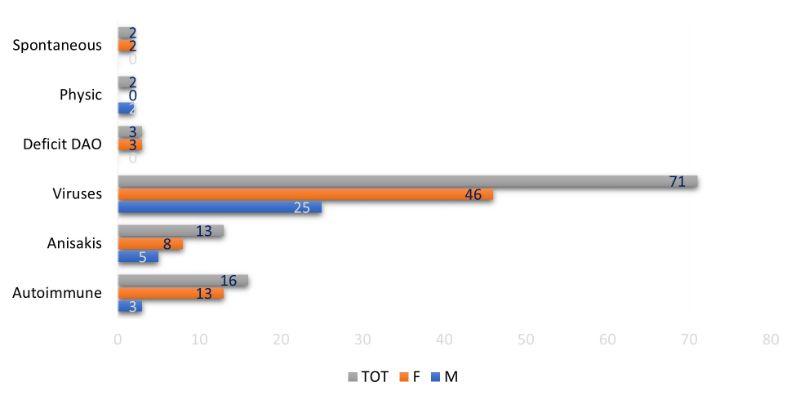
Fig 3 Chronic urticaria’s aetiology
Regarding the viral diagnostic work-up, the aVGt/TR in patients was significantly higher (more than 10-fold) compared to healthy controls (Fig. 4); the statistical analysis of the contingency table showed very high significance (χ² = 59.17, p < 0.0005, Odds Ratio = 11.95, 99% confidence interval) (Tab.1).
Tab.1 Contingency table (aVGt/T Rt=antivirus IgG titers/Threshold Ratio)
|
aVGt/T Rt |
Patients |
Controls |
TOT |
|
>10 |
71 |
17 |
88 |
|
<10 |
29 |
83 |
112 |
|
TOT |
100 |
100 |
200 |
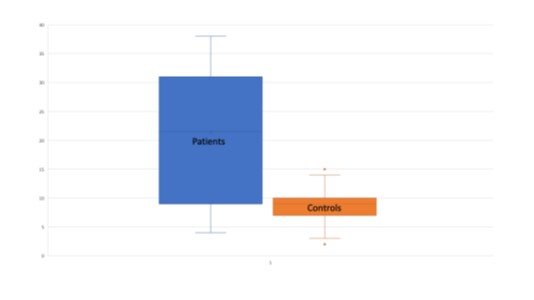
Fig.4: Blood antivirus IgG titers/Threshold Ratio in CU Patients and Controls + SD.
Therefore, an IgG antiviral titre was considered a marker of recent viral reinfection/reactivation when the value exceeded approximately ten times the threshold value (8,19). Conversely, an aVGt/TR lower than 10‑fold was attributed to immunological memory from past infection.
Therapies were aimed at treating symptoms (second‑generation antihistamines, anti‑leukotrienes, antioxidants, and low‑dose corticosteroids only when necessary and for periods not exceeding 2–4 weeks), treating the identified aetiology (antivirals, anthelmintics, synthetic DAO, and small ozonized autohemotherapy), and managing relevant comorbidities (restoration of intestinal microbiota and treatment of underlying autoimmune diseases).
The patient‑reported outcomes (Fig. 5) were:
A) Healing 74%;
B) Improvement 20%;
C) Unchanged 6%.
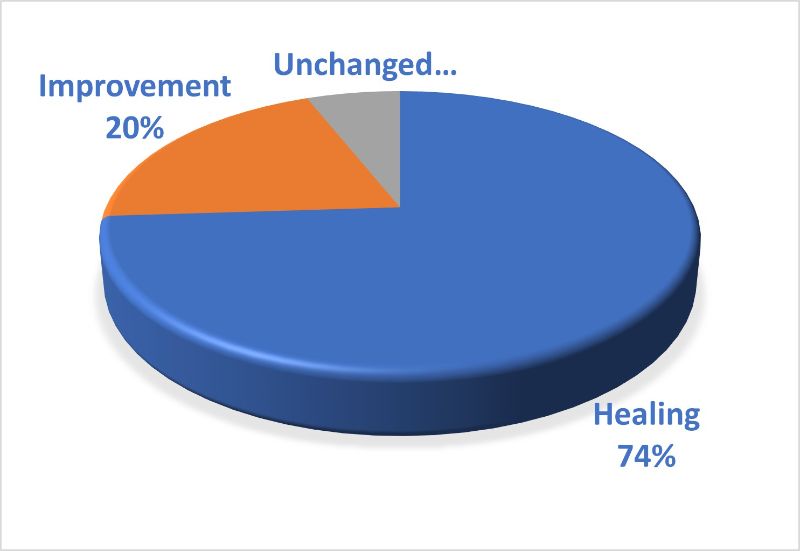
Fig.5 Global clinical outcomes in 100 patients suffering from Chronic Urticaria.
Discussion
Chronic urticaria can be triggered by multiple factors that converge on a single effector responsible for symptoms: the cutaneous mast cell. Mast cells stem from bone‑marrow‑derived precursors that spread throughout the body; while their cytologic features are originally similar, their phenotype and mediator content diverge according to tissue localization (27). In humans, mast cells resident in mucous membranes lining hollow organs exposed to the external environment (airways, gut, and genitourinary tract) contain mainly tryptase (MCT), whereas those resident in the skin and connective tissues of internal organs also contain chymase (MCTC).
These cells can be detected by optical microscopy using standardized cytological methods after classic May–Grünwald–Giemsa staining (60). Vasodilation and neovessel proliferation with increased inflammation are induced by serine proteases, vascular endothelial growth factor (VEGF), other mitogens, and histamine released by mast cells (52,54).
Many triggers can unleash mast cell degranulation (Fig. 1): IgE–allergen immune complexes leading to classic type I reactions; IgE–autoantigen immune complexes (22,55); IgG–pathogen immune complexes; IgG–autoantigen immune complexes; viruses; and bacterial products such as peptidoglycans and lipoteichoic acid. These triggers engage different membrane structures, including receptors for alarmins (IL‑33, Thymic Stromal Lymphopoietin -TSLP-) (63), Fc receptors, and pattern‑recognition receptors (PAMPs and TLRs): FcγR for IgG immune complexes, FcεRI for IgE immune complexes, TSLPR, ST2 for IL‑33, TLR2 and TLR4 for bacterial peptidoglycans and lipoteichoic acid, TLR3 for dsRNA viruses (e.g., rotaviruses), TLR7/8 for ssRNA viruses (e.g., COVID‑19 and DENV), and TLR9 for DNA viruses (e.g., EBV, HSV, CMV, and VZV) (27).
Amplification of triggering effects may arise from keratinocytes stimulated by invading viruses and/or bacteria. Skin epithelial cells then release emergency signals such as alarmins (TSLP, IL‑33) and growth factors such as SCF. SCF engages the mast cell c‑kit receptor, inducing proliferation and degranulation, while IL‑33 increases expression of membrane ICAM‑1. ICAM‑1 promotes intercellular adhesion and interactions; at mucosal surfaces it can also serve as a gateway for human rhinovirus (HRV), potentially increasing invasion (30). Increased production of VEGF enhances angiogenesis and local inflammation and has been associated with adverse prognostic features in oncology (53).
Viruses can also directly infect target cells, including mast cells. Regarding viral diagnostic work‑up, it should be considered that viruses live and bud within parasitized cells, and most of their life cycle occurs in the cytoplasm. Therefore, viraemia may be transient, and serum polymerase chain reaction testing may be uninformative if sampling does not coincide with viral shedding into the bloodstream. Moreover, viruses may persist within circulating cells (e.g., lymphocytes), which can act as carriers to reach target tissues, acting as a sort of Trojan horse. Some reticuloendothelial‑system‑rich organs can act as reservoirs for virions; cytoplasmic proteolytic enzymes enable the partial digestion of viral capsid proteins, leading to viral transcription (61).
Another route used by viruses to invade cells is via neural axons, as in the case of shingles; the entry point in this case is the acetylcholine receptor (AChR), the same target of neurotoxins contained in some snake venoms. Viruses can cross this gateway bidirectionally, invading both neuronal axons and the cells at the neuromuscular junction, including mast cells.
In the skin, mast cells are located in the papillary dermis, near nerve fibres and around dermal blood vessels (62); therefore, they could plausibly contribute to local viral persistence and immune activation in chronic urticaria. Circulating antibodies against viruses are commonly detectable in blood, and their titre reflects infection history. After first exposure, IgM antibodies are produced and then replaced after about two weeks by IgG. Reinfections or reactivations may manifest mainly as an IgG increase. In immunocompetent subjects, memory T and B cells persist for about ten years; if reinfection occurs within this period, IgG synthesis can be faster and higher (8,13,18,26), and IgM synthesis does not occur. Therefore, a virus‑specific IgG titre less than about 10 times the threshold may reflect past immunological memory, whereas higher titres were interpreted as a marker of reinfection/reactivation, with greater values suggesting more recent infection. In our cohort, the ratio of virus‑specific IgG titres to threshold was significantly higher (more than 10‑fold) in patients with chronic viral urticaria (71%) than in healthy controls (17%). Monitoring the antiviral IgG titre/threshold ratio over time can help clinicians assess infection behaviour: a decreasing ratio indicates past infection, whereas a rising ratio suggests ongoing activity.
Regarding therapy, treating viruses may require antivirals, antioxidants, immunomodulators (e.g., lysozyme), and correction of factors associated with susceptibility to infections (e.g., restoration of intestinal microbiota in dysbiosis, correction of nutritional deficiencies, management of comorbidities). A case report described the efficacy of small ozonized autohemotherapy (intramuscular injection of the patient’s blood exposed to an oxygen–ozone mixture), which resolved two cases of chronic urticaria associated with EBV and HSV1 (16). In this approach, exposure of blood to oxygen–ozone may inactivate pathogens because ozone has documented bactericidal and virucidal activity (57,58,59); the intramuscular injection of ozonated autologous blood, when performed according to validated protocols, may act as a form of autovaccine (64).
For gut parasites, including Anisakis, anthelmintic drugs such as mebendazole or albendazole are appropriate; numerous complications have been described in untreated anisakiasis (14,31,33–35), including intestinal bleeding (39), gastric cancer (36,37), intestinal ischemia (40), extraintestinal larval migration with respiratory manifestations (41), eosinophilic esophagitis (32), and hypereosinophilic syndrome (38).
After excluding autoimmune diseases, gut parasites, or viral causes, DAO deficiency may be detected. DAO is produced by intestinal villus epithelial cells and catabolizes histamine released by intestinal mast cells and enterochromaffin cells during digestion. In primary or secondary deficiency, circulating histamine may persist longer, potentially contributing to chronic urticaria; this clinical picture is often referred to as histamine intolerance (7,42,43,45). In such cases, it is appropriate to administer synthetic DAO and prescribe a low histamine‑releasing diet. A randomized double‑blind clinical trial showed synthetic DAO to be helpful in patients with chronic urticaria (45).
Moreover, failure to maintain the cold chain during storage of frozen foods (e.g., fish) can lead to a histaminergic syndrome mimicking anaphylaxis (Scombroid Fish Syndrome) (56), due to high histamine content resulting from bacterial histidine decarboxylase activity in improperly stored food.
Conclusions
In conclusion, mast cells in the skin and at mucosal surfaces act as advanced immune sentinels in barrier tissues. They are equipped with a wide range of membrane receptors for environmental cues and contain preformed mediators ready for release, while also being able to synthesize new ones when needed. The breadth of receptors expressed by mast cells can make them targets of pathogen‑associated stimuli; in our cohort, viral infections were frequently associated with chronic urticaria. While host‑protective, mast‑cell‑mediated responses may sometimes damage host tissues and contribute to trigger conditions such as chronic urticaria.
A structured diagnostic work‑up combined with careful assessment of triggers able to activate skin mast cells allowed identification of aetiology in 95% of cases; targeted therapies achieved healing in three‑quarters of patients and improvement in one‑fifth. Only 6% of patients did not respond effectively; thus, the term “spontaneous” should arguably be reserved for these cases of chronic urticaria. Hospitalization and the use of biologic drugs limited to such refractory patients could involve significant cost savings for the National Health Service.
Compliance with Ethical Standards statements
The Author declares that there is no funding and no conflict of interest associated with this manuscript.
Impact Statement
Chronic spontaneous urticaria is often treated with expensive biologic drugs. By performing an appropriate diagnostic work‑up and applying targeted treatments, it is possible to achieve substantial public cost savings for the National Health Service.