International Journal of Integrative and Complementary Medicine
OPEN ACCESS | Volume 2 - Issue 1 - 2026
ISSN No: 3070-4146 | Journal DOI: 10.61148/3070-4146/IJICM
F. A. Adewumi1*, M. O. Lateef2, M. O. Oseni3, A. I. Airaodion4, O. A. Oseni5
1Department of Medical Laboratory Science, Faculty of Basic Medical Sciences, College of Medicine, Ekiti State University, Ado- Ekiti, Nigeria.
2Department of Science Laboratory Technology, Faculty of Science, Ekiti State University, Ado-Ekiti, Nigeria.
3Department of Chemistry, Faculty of Physical Science, Federal University, Oye-Ekiti, Ekiti State, Nigeria.
4Department of Biochemistry, Faculty of Natural and Applied Sciences, Lead City University, Ibadan, Oyo State, Nigeria.
5Department of Medical Biochemistry, Faculty of Basic Medical Sciences, College of Medicine, Ekiti State University, Ado- Ekiti, Nigeria.
*Corresponding author: F. A. Adewumi, Department of Medical Laboratory Science, Faculty of Basic Medical Sciences, College of Medicine, Ekiti State University, Ado- Ekiti, Nigeria.
Received: February 06, 2026 | Accepted: February 13, 2026 | Published: February 18, 2026
Citation: F. A. Adewumi, M. O. Lateef, M. O. Oseni, A. I. Airaodion, O. A. Oseni., (2026) “Integrated Evaluation of Bauhinia purpurea Leaf: Antimicrobial Efficacy and Antioxidant Potential of Leaf Extracts and Green Synthesized Silver Nanoparticles.” International Journal of Integrative and Complementary Medicine, 2(1). DOI: 10.61148/ 10.61148/IJICM/013.
Copyright: © 2026 F. A. Adewumi. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Background: Bauhinia purpurea is a leguminous tree that features prominently in traditional diets and medicine across many tropical regions. Although it is widely used, studies that jointly examine its nutritional value, phytochemical makeup, antioxidant performance, and antimicrobial effects, especially with direct comparisons between crude extracts and biosynthesized silver nanoparticles, remain scarce.
Methods: Fresh leaves of B. purpurea were harvested in the early hours of March 10, 2025, from naturally occurring stands in Ado-Ekiti, Ekiti State, Nigeria. The plant material was taxonomically authenticated and evaluated for proximate composition, mineral content, and antinutritional factors. Qualitative phytochemical screening was carried out, alongside quantitative estimation of total phenolics and flavonoids. The antioxidant activities of aqueous and ethanol extracts were tested in vitro using DPPH, FRAP, nitric oxide, and TBAR assays at a concentration of 100 µg/mL. Antimicrobial activity of the aqueous extract and green-synthesized silver nanoparticles (AgNPs) was assessed against Escherichia coli, Salmonella typhi, Staphylococcus aureus, and Pseudomonas aeruginosa.
Results: Proximate analysis showed that the leaves were rich in carbohydrates (48.57 ± 0.03%), with notable protein (17.74 ± 0.05%) and fibre (10.20 ± 0.02%) contents, alongside low moisture (7.40 ± 0.01%) and moderate ash levels (11.30 ± 0.03%). Antinutritional components such as oxalate, phytate, tannins, and trypsin inhibitors occurred at low concentrations, indicating minimal interference with nutrient utilization. Beneficial minerals, including potassium, sodium, calcium, magnesium, and iron, were present in appreciable amounts. Phytochemical analysis confirmed the presence of diverse bioactive compounds, notably phenolics, flavonoids, alkaloids, terpenoids, and glycosides. While the aqueous extract was richer in hydrophilic antioxidants, the ethanol extract displayed stronger overall antioxidant activity across all four assays. In antimicrobial tests, the crude aqueous extract inhibited E. coli, S. typhi, and S. aureus but showed no activity against P. aeruginosa. In contrast, the AgNPs exhibited enhanced inhibitory effects against all tested organisms.
Conclusion: Leaves of Bauhinia purpurea offer a valuable combination of nutrients and bioactive phytochemicals, with marked antioxidant and inherent antimicrobial properties that are further amplified through green synthesis of silver nanoparticles. These findings highlight the plant’s promise as a functional food resource and as a basis for developing plant-derived nanotherapeutic agents. Blending traditional phytochemical insights with modern nanotechnology opens new avenues for applications in food science and biomedicine.
Bauhinia purpurea, phytochemical screening, antioxidant activity, antimicrobial efficacy, silver nanoparticles, green synthesis
Medicinal plants have played a central role in human healthcare for centuries, forming the backbone of traditional medicine and serving as critical sources for modern drug discovery. Their therapeutic relevance is largely attributed to diverse secondary metabolites such as flavonoids, terpenoids, alkaloids, and phenolic compounds, which exhibit antimicrobial, antioxidant, anti-inflammatory, and anticancer activities [1].
One such plant is Bauhinia purpurea L., commonly known as the Purple Orchid Tree, a member of the Fabaceae family widely distributed across tropical regions. It is extensively used in ethnomedicine for the treatment of wounds, ulcers, diabetes, infections, liver disorders, and other ailments. Phytochemicals isolated from B. purpurea have demonstrated antimalarial, antimycobacterial, antifungal, anti-inflammatory, and cytotoxic effects, highlighting its broad therapeutic potential [2].
The integration of plant science with nanotechnology has opened new research pathways, particularly through green synthesis techniques. In this approach, plant extracts act as both reducing and stabilizing agents in the production of metal nanoparticles, eliminating the need for toxic chemicals. These eco-friendly nanoparticles often display enhanced biological activities and biocompatibility compared to crude plant extracts [1,3]. For example, silver and gold nanoparticles synthesized using B. purpurea leaf extracts have shown remarkable antimicrobial, antioxidant, and anticancer properties in vitro [3].
Despite the extensive traditional use of B. purpurea and isolated pharmacological reports, few studies have combined phytochemical profiling, green nanoparticle synthesis, and bioactivity evaluation in a single framework. An integrated approach is therefore essential to advance this plant from traditional application to evidence-based biomedical development. This study was designed to provide a comprehensive evaluation of Bauhinia purpurea, combining phytochemical analysis, environmentally friendly nanoparticle synthesis, and biological activity assessment to explore its potential therapeutic relevance.
RESEARCH METHODOLOGY
Plant Material Collection and Taxonomic Authentication
Fresh leaves of Bauhinia purpurea (Orchid plant) were collected on March 10, 2025, in the early morning from naturally growing stands in Ado-Ekiti, Ekiti State, Nigeria. Early morning collection was chosen to minimize moisture loss and preserve heat-sensitive phytochemicals that may degrade under strong sunlight or elevated temperatures. Proper handling of the samples during collection ensured that primary bioactive constituents (e.g., flavonoids, phenolics, alkaloids, and saponins) remained intact for subsequent analysis. The species identity was authenticated at the Herbarium of the Department of Plant Science, Faculty of Science, Ekiti State University, Ado-Ekiti, Nigeria, where a voucher specimen was prepared and deposited for future reference.
Preparation of Aqueous Leaf Extract
Leaves were thoroughly rinsed under running tap water to remove surface dust and debris, then air-dried at ambient temperature until constant weight was achieved. The dried leaves were pulverized into fine powder using a sterile mortar and pestle to increase extraction efficiency. One hundred grams (100 g) of the powdered leaves were weighed and transferred into a clean extraction beaker. One litre (1 L) of distilled water was added to the powder, and the mixture was stirred to ensure uniform dispersion. The suspension was heated on a heating mantle at 70–80 °C for 2 h to facilitate the release of soluble phytochemicals into the solvent, a strategy commonly employed in plant extraction protocols to improve the yield of antioxidant constituents (e.g., phenolics and flavonoids) known to contribute to bioactivity in aqueous extracts [4]. After cooling to room temperature, the mixture was filtered through a fine cloth, and the resulting filtrate was collected into clean, labelled containers. The aqueous extract was stored at 4 °C until further analysis to maintain stability.
Qualitative Phytochemical Screening
The aqueous leaf extract was subjected to qualitative screening to detect major classes of secondary metabolites, including flavonoids, phenolic compounds, alkaloids, saponins, and steroids. Standard chemical tests, which involve reagent reactions producing characteristic colour changes or precipitates, were used to confirm the presence of these phytochemicals, as previously demonstrated in aqueous extracts of Bauhinia purpurea where multiple phytochemicals were identified in qualitative analyses [5,6].
In Vitro Antioxidant Assays
DPPH Free Radical Scavenging Assay
The 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging assay was used to evaluate the antioxidant activity of the extract. Different extract concentrations (e.g., 20, 40, and 80 mg/mL) were prepared in clean test tubes. One millilitre (1 mL) of each concentration was mixed with 1 mL of 0.1 mM methanolic DPPH solution. The mixtures were vortexed and incubated in the dark at room temperature for 30 min to allow interaction between antioxidants in the extract and the DPPH radical. After incubation, absorbance was measured at ~516 nm (the characteristic absorption wavelength of DPPH radical) using a UV-visible spectrophotometer. A significant reduction in absorbance relative to a control indicates effective free radical scavenging, as previously reported for aqueous leaf extracts of Bauhinia purpurea with notable DPPH scavenging activity in published studies [7,8].
Green Synthesis of Metal Nanoparticles
Green synthesis of metal nanoparticles (e.g., silver, copper, zinc) was conducted using the aqueous leaf extract as a reducing and stabilizing agent, following plant-mediated synthesis protocols widely described in nanobiotechnology literature. In this approach, aqueous solutions of metal salts (such as AgNO₃ for silver nanoparticles) are mixed with plant extract under controlled stirring and temperature conditions, allowing bioactive phytochemicals to reduce metal ions and cap the resulting nanoparticles. Such eco-friendly synthesis methods are known to yield nanoparticles with bioactive surfaces suitable for downstream biological evaluation, including antioxidant and antimicrobial assays [3].
Antimicrobial Evaluation of Synthesized Nanoparticles
The antimicrobial activities of the green-synthesized nanoparticles were assessed against selected pathogenic bacterial strains using standard microbiological assays such as the agar well diffusion method. Test organisms were cultured on appropriate media, and standardized inocula were spread onto agar plates. Wells loaded with nanoparticle suspensions were incubated, and zones of inhibition were measured to evaluate bacteriostatic and bactericidal effects relative to controls.
Additional Antioxidant Assessments
To complement the DPPH assay, total phenolic content and flavonoid content of the extract were estimated using spectrophotometric assays with relevant standards. These complementary assays provide quantitative insights into specific antioxidant components contributing to overall activity, which is essential in studies exploring extracts as both biological agents and reducing agents for green nanoparticle synthesis [4].
RESULTS
The proximate composition of Bauhinia purpurea leaf is presented in Table 1, showing moisture (7.40 ± 0.01%), ash (11.30 ± 0.03%), crude fat (4.79 ± 0.05%), crude fibre (10.20 ± 0.02%), crude protein (17.74 ± 0.05%), and carbohydrate (48.57 ± 0.03%). The antinutrient profile in Table 2 indicates oxalate (3.16 ± 0.01 mg/100 g), phytate (11.52 ± 0.02 mg/100 g), tannin (16.55 ± 0.00 mg/100 g), and trypsin inhibitor (21.00 ± 0.39%).
Mineral composition is summarized in Table 3, with potassium (4.52 ± 0.02%), sodium (2.53 ± 0.04%), calcium (2.13 ± 0.01%), copper (0.93 ± 0.00%), chromium (0.11 ± 0.00%), magnesium (0.98 ± 0.03%), iron (2.68 ± 0.00%), manganese (0.29 ± 0.00%), and lead (0.10 ± 0.00%).
Phytochemical screening of the aqueous extract is shown in Table 4, indicating the presence of saponins, phenolics, flavonoids, anthraquinones, terpenoids, and cardiac glycosides, while steroids were not detected.
Table 5 presents the flavonoid and phenolic contents of ethanol and aqueous extracts at 100 µg/mL. The ethanol extract showed flavonoids of 16.54 ± 0.47 mg AAE/g and phenolics of 13.01 ± 0.28 mg GAE/g, while the aqueous extract showed flavonoids of 21.13 ± 0.01 mg AAE/g and phenolics of 32.56 ± 0.11 mg GAE/g.
The antioxidant activities of the aqueous extract at 100 µg/mL are shown in Table 6, with DPPH (48.39 ± 0.04), FRAP (57.15 ± 0.57), nitric oxide scavenging (65.15 ± 0.85), and TBAR (22.23 ± 0.84). Table 7 summarizes the antioxidant activities of the ethanol extract at 100 µg/mL, showing DPPH (76.54 ± 0.47), FRAP (74.93 ± 0.13), nitric oxide scavenging (88.13 ± 1.56), and TBAR (26.30 ± 0.11).
The antimicrobial activity of the aqueous extract and silver nanoparticles is presented in Table 8. At 100 mg/mL, the aqueous extract produced inhibition zones of 3.7 mm against E. coli, 2.8 mm against S. typhi, and 1.3 mm against S. aureus, with no activity against P. aeruginosa. At 50 mg/mL, inhibition zones were 2.9 mm, 2.2 mm, and 1.0 mm against E. coli, S. typhi, and S. aureus, respectively. The silver nanoparticles at 100 mg/mL showed inhibition zones of 5.3 mm (E. coli), 4.1 mm (S. typhi), 3.0 mm (S. aureus), and 1.2 mm (P. aeruginosa).
Table 1: Proximate Composition of Bauhinia purpurea Leaf
|
Parameter |
Value (%) |
|
Moisture Content |
7.40 ± 0.01ᵃ |
|
Ash Content |
11.30 ± 0.03ᵇ |
|
Crude Fat |
4.79 ± 0.05ᶜ |
|
Crude Fiber |
10.20 ± 0.02ᵇ |
|
Crude Protein |
17.74 ± 0.05ᵈ |
|
Carbohydrate (CHO) |
48.57 ± 0.03ᵉ |
Table 2: Antinutrient Composition of Bauhinia purpurea Leaf
|
Antinutrient Parameter |
Value (%) / mg/100 g |
|
Oxalate (mg/100 g) |
3.16 ± 0.01 |
|
Phytate (mg/100 g) |
11.52 ± 0.02 |
|
Tannin (mg/100 g) |
16.55 ± 0.00 |
|
Trypsin Inhibitor (%) |
21.00 ± 0.39 |
Table 3: Mineral Composition of Bauhinia purpurea leaf
PARAMETERS VALUE%
K 4.52±0.02
Na 2.53±0.04
Ca 2.13±0.01
Cu 0.93±0.00
Cr 0.11±0.00
Mg 0.98±0.03
Fe 2.68±0.00
Mn 0.29±0.00
Pb 0.10±0.00
Table 4: Phytochemical screening of the Aqueous plant extracts (Bauhinia purpurea)
Parameters Value (%)
Saponin Test +
Phenolic Test ++++
Wagner’s Test ++++
Anthraquinone Test +
Terpenoid Test ++++
Keller-killani Test +++
Lieberman’s Test -
Table 5: Antioxidant potentials of Aqueous Plant extracts of Bauhinia purpurea leaf
|
Conc ((????g/mL)) |
ETHANOL |
AQUEOUS |
||
|
FLAVONOIDS (mg AAE/g) |
PHENOLICS (mg GAE/g) |
FLAVONOIDS (mg AAE/g) |
PHENOLICS (mg GAE/g) |
|
|
100 |
16.54±0.47 |
13.01±0.28 |
21.13±0.01 |
32.56±0.11 |
Table 6: Antioxidant potentials of aqueous plant’s extracts of Bauhinia purpurea leaf
|
Conc (????g/mL) |
AQUEOUS Antioxidant |
|
||
|
DPPH |
FRAP |
NO |
TBAR |
|
|
100 |
48.39±0.04 |
57.15±0.57 |
65.15±0.85 |
22.23±0.84 |
Table 7: Antioxidant potentials of ethanol plant’s extracts of Bauhinia purpurea leaf
|
Conc (????g/mL) |
ETHANOL Antioxidant |
|
||
|
DPPH |
FRAP |
NO |
TBAR |
|
|
100 |
76.54±0.47 |
74.93±0.13 |
88.13±1.56 |
26.30±0.11 |
DPPH – 2,2‑diphenyl‑1‑picrylhydrazyl, FRAP – Ferric Reducing Antioxidant Power. NO – Nitric Oxide scavenging assay, TBAR – Thiobarbituric Acid Reactive Substances,
Table 8: Antimicrobial activity of Aqueous extract and Silver Nanoparticle of Bauhinia purpurea leaf on some selected bacteria
|
Sample |
Concentration (mg/ml) |
E. coli |
S. typhi |
S. aureus |
P. aeruginosa |
|
Aqueous extract |
100 |
3.7 |
2.8 |
1.3 |
- |
|
50 |
2.9 |
2.2 |
1.0 |
- |
|
|
Silver Nanoparticles (AgNP) |
100 |
5.3 |
4.1 |
3.0 |
1.2 |
DISCUSSION
This work provides a holistic appraisal of Bauhinia purpurea leaves by combining nutritional profiling with phytochemical screening, antioxidant evaluation, and antimicrobial testing of both crude extracts and biosynthesized silver nanoparticles (AgNPs). The findings show that the plant is not only nutritionally valuable but also pharmacologically promising.
The proximate data (Table 1) indicate that B. purpurea leaves are particularly rich in carbohydrates (48.57 ± 0.03%), proteins (17.74 ± 0.05%), and dietary fibre (10.20 ± 0.02%), while having a relatively low moisture content (7.40 ± 0.01%). This combination supports both energy provision and storage stability. Comparable trends have been reported for edible leafy vegetables, which are known to enhance digestion and contribute substantially to daily energy intake because of their carbohydrate and fibre content [9]. The moderate levels of crude fat (4.79 ± 0.05%) and ash (11.30 ± 0.03%) further point to the presence of essential fatty acids and minerals that are important for membrane integrity, cellular signalling, and micronutrient balance.
The antinutrient contents (Table 2), including oxalate, phytate, tannins, and trypsin inhibitor activity, were generally low and within acceptable dietary limits. Such levels are unlikely to pose major risks related to mineral binding or protein digestibility when the leaves are eaten as part of a mixed diet or after common processing steps. Interestingly, controlled quantities of tannins and protease inhibitors have been linked with antioxidant and antimicrobial functions in plants, suggesting that these compounds may contribute to the observed bioactivities rather than detract from nutritional quality.
The mineral analysis (Table 3) confirmed that the leaves contain useful amounts of macro elements such as potassium, sodium, calcium, and magnesium, as well as trace elements including iron, copper, manganese, and chromium. These minerals play central roles in electrolyte regulation, bone health, enzymatic reactions, and haemoglobin synthesis. Of particular importance is the very low level of lead detected, which supports the safety of the leaves for dietary and medicinal applications.
Qualitative screening (Table 4) revealed a wide range of secondary metabolites such as phenolics, flavonoids, alkaloids, terpenoids, saponins, and cardiac glycosides. This profile is consistent with reports that members of the Bauhinia genus are rich in bioactive compounds with diverse pharmacological effects, including antioxidant, anti-inflammatory, and antimicrobial actions [10]. These phytochemicals form the biochemical basis for many of the functional properties observed in this study.
Quantitative assays (Table 5) showed that the aqueous extract contained higher total phenolic and flavonoid contents than the ethanol extract, highlighting the efficiency of water in extracting hydrophilic antioxidants. However, functional antioxidant tests (Tables 6 and 7) demonstrated that the ethanol extract exhibited stronger activity across several mechanisms, including DPPH radical scavenging, ferric reducing power, nitric oxide inhibition, and suppression of lipid peroxidation. This likely reflects ethanol’s ability to solubilize both polar and moderately non-polar antioxidant compounds, a pattern also reported in studies on B. purpurea and related green nanoparticle systems [11].
The antimicrobial assays showed that the crude aqueous extract inhibited Escherichia coli, Salmonella typhi, and Staphylococcus aureus in a concentration-dependent manner, but had little effect on Pseudomonas aeruginosa. Similar observations have been made in earlier work, where aqueous extracts of B. purpurea displayed weaker activity than organic solvent extracts, which were more effective against both Gram-positive and some Gram-negative bacteria [12]. This difference is often attributed to the higher solubility of antimicrobial phytochemicals in polar organic solvents.
In contrast, the biosynthesized AgNPs demonstrated markedly enhanced antibacterial activity against all tested strains, including P. aeruginosa. At 100 mg/mL, the AgNPs produced larger zones of inhibition than the crude extracts. This agrees with previous studies showing that plant-mediated AgNPs possess broad-spectrum antibacterial effects that exceed those of the parent extracts because of synergistic mechanisms such as membrane disruption, increased reactive oxygen species generation, and interference with DNA replication and cellular respiration [3]. Similar results have been reported for AgNPs synthesized using B. purpurea and other Bauhinia species, reinforcing the idea that nanoscale formulation amplifies antimicrobial potency in this genus [13].
CONCLUSION
The strong nutritional profile, balanced antinutrient levels, rich mineral and phytochemical composition, robust antioxidant activity, and especially the enhanced antimicrobial efficacy of the AgNPs position B. purpurea leaves as a promising functional food and a source of natural antioxidants and alternative antimicrobial agents. The clear synergy between traditional phytochemicals and nanotechnology supports further investigation of this plant for nutraceutical and biomedical applications, including mechanistic studies and formulation development.
APPENDIX
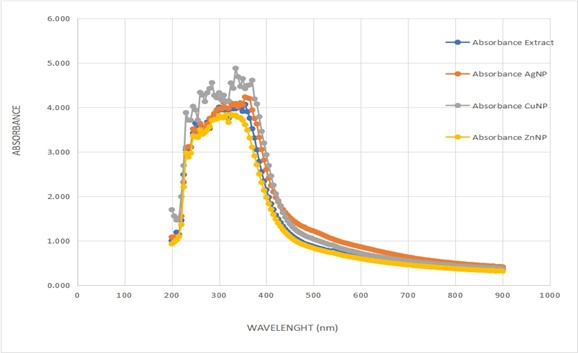
Figure 1.0: The Wavelength scanning of Bauhinia purpurea aqueous extract with metallic nanoparticles
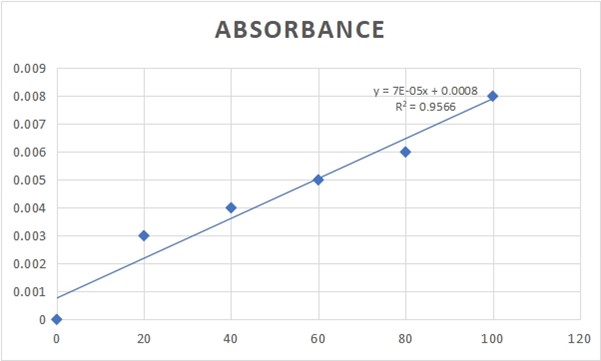
Figure 2.0: FLAVANOID CONCENTRATION AND ABSORBANCE GRAPH
%inhibition of nitric oxide (NO) radical = [(Ai – At)/Ai] *100
Ai =Absorbance of control
At =Absorbance of sample
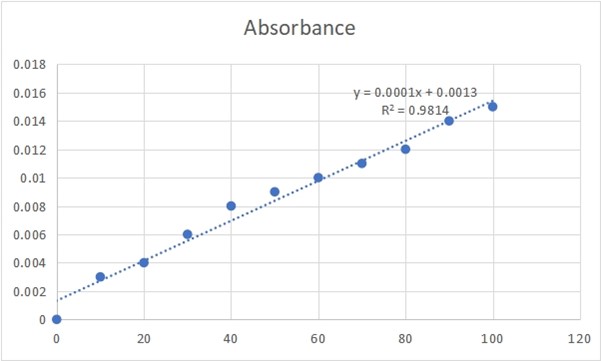
Figure 3.0: PHENOL CONCENTRATION AND ABSORBANCE GRAPH
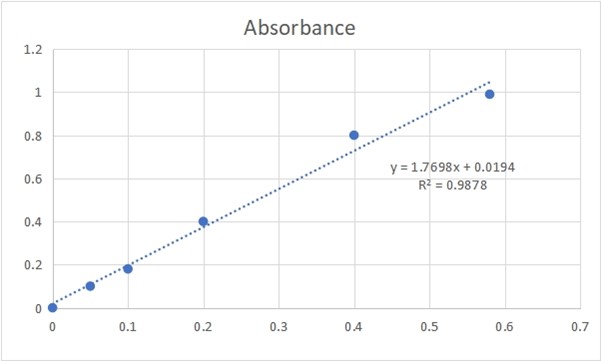
Figure 4.0: ASCORBIC ACID CONCENTRATION AND ABSORBANCE GRAPH
Sample 1D: Red frangipani (plumeria rubra) plant aqueous extract
Resolution :8
Full Scale 42243
Method: Transmittance Method
Sample Spectrum
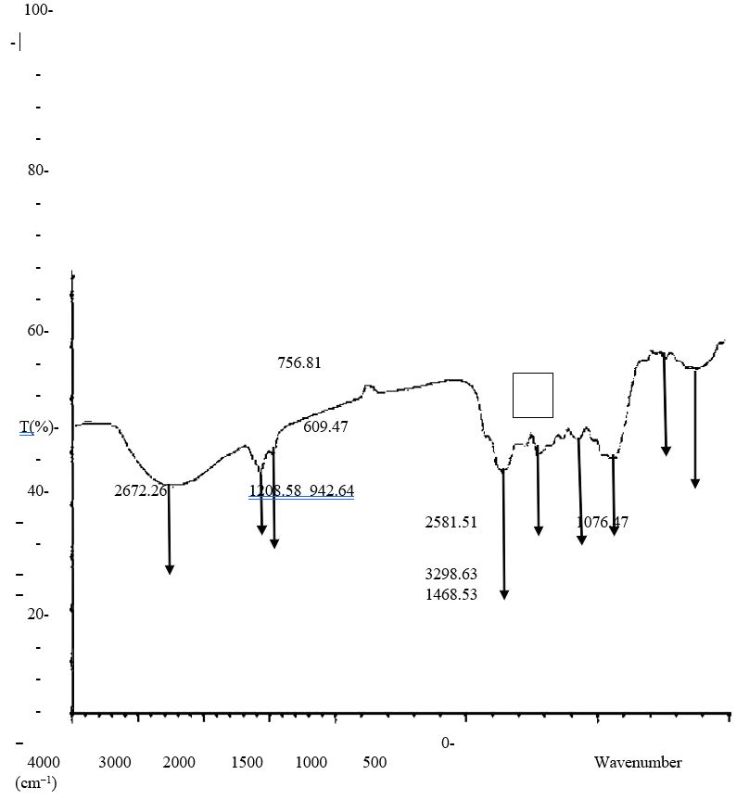
Figure 5.0. FTIR characterization of Orchid plant (Bauhinia purpurea) aqueous extract
Sample Scan: 200 scans
Resolution :8
Method: Transmittance Method
Sample Spectrum
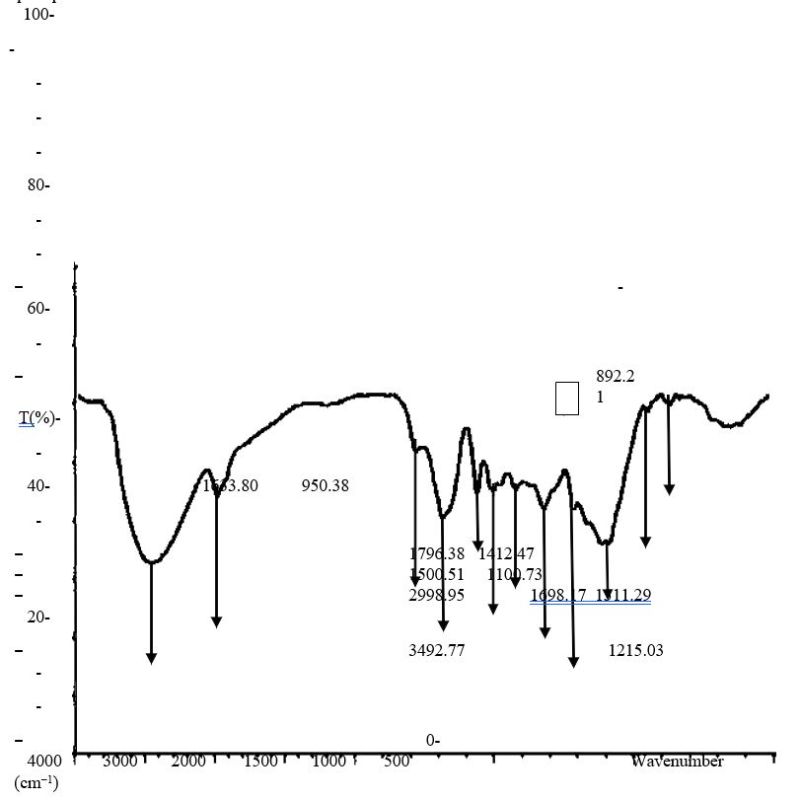
Figure 6.0. FTIR characterization of Orchid plant (Bauhinia purpurea) AgNP
Sample Scan: 200 scans
Resolution :8
Method: Transmittance Method
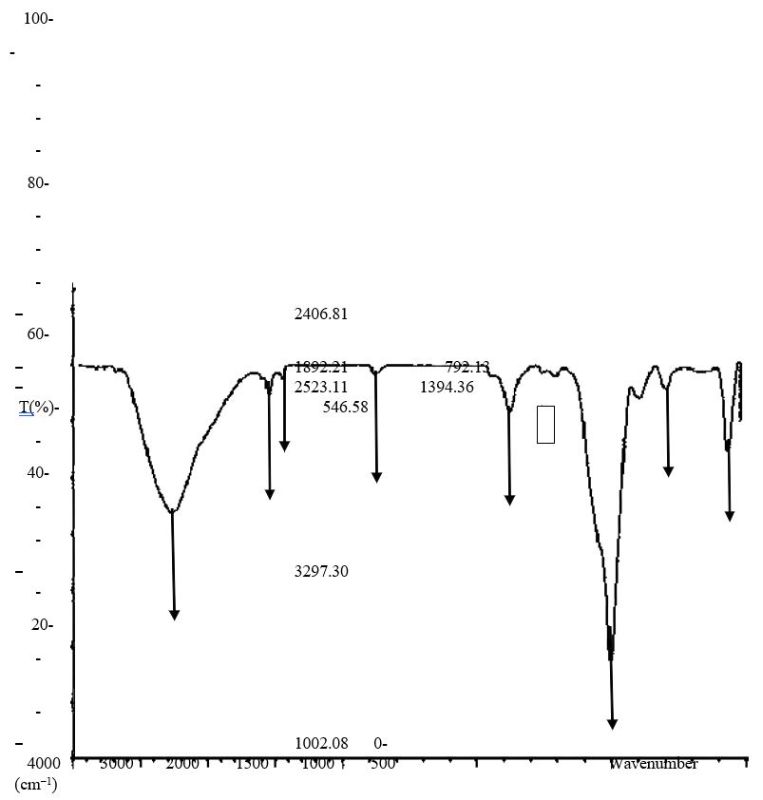
Figure 7.0. FTIR characterization of Orchid plant (Bauhinia purpurea) ZnNP
Sample Scan: 200 scans
Resolution :8
Method: Transmittance Method
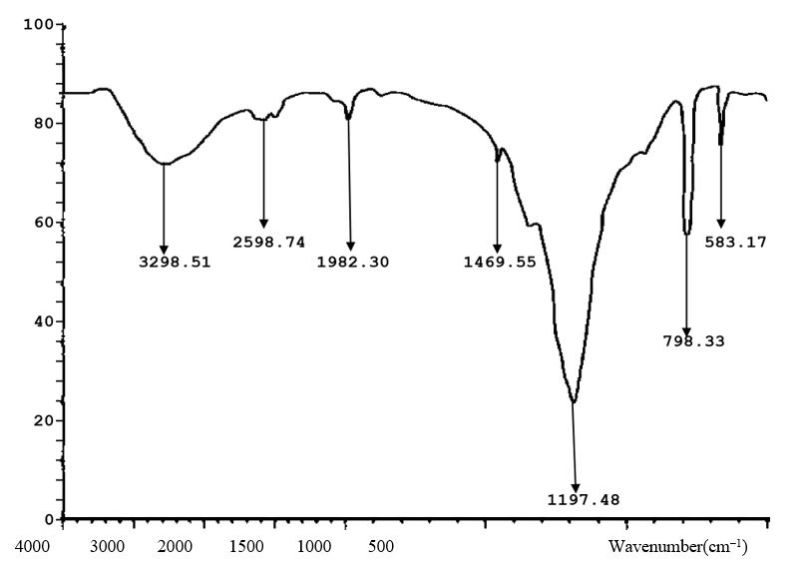
Figure 8.0. FTIR characterization of Orchid plant (Bauhinia purpurea) CuNP
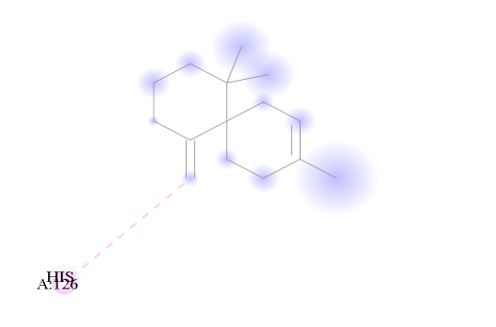
Figure 9.0: 2D Representation showing the binding of Spiro[5.5]undec-2-ene, 3,7,7-trimethyl-11-methylene-, (-)- as the top performer ligand against the target macromolecule (DPP4) carried out with the aid of BIOVIA Discovery Studio.
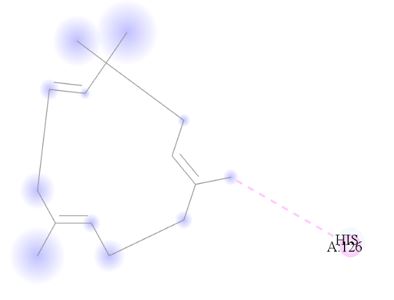
Figure 10: 2D Representation showing the binding of Humulene as the top performer ligand against the target macro molecule (DPP4) carried out with the aid of BIOVIA Discovery Studio.
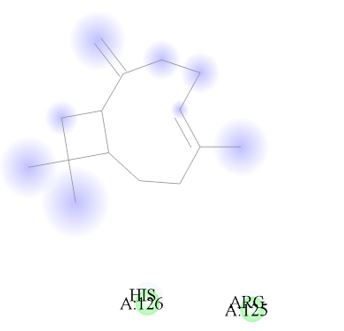
Figure 11: 2D Representation showing the binding of Caryophyllene as the top performer ligand against the target macro molecule (DPP4) carried out with the aid of BIOVIA Discovery Studio.
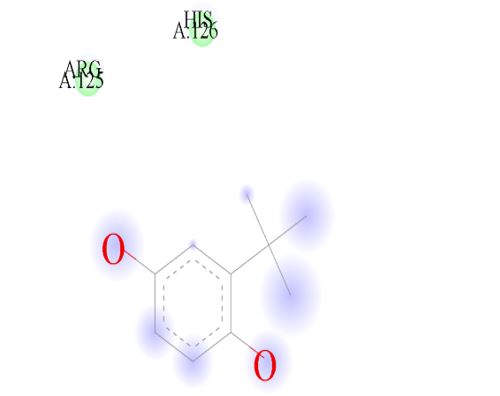
Figure 12: 2D Representation showing the binding of t-Butylhydroquinone as the top performer ligand against the target macro molecule (DPP4) carried out with the aid of BIOVIA Discovery Studio
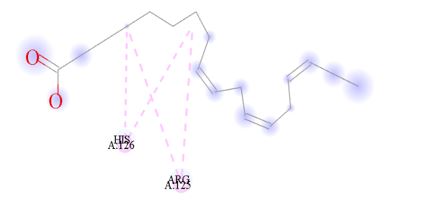
Figure 13: 2D Representation showing the binding of 9,12,15-Octadecatrienoic acid, (Z,Z,Z)- as the top performer ligand against the target macro molecule (DPP4) carried out with the aid of BIOVIA Discovery Studio.
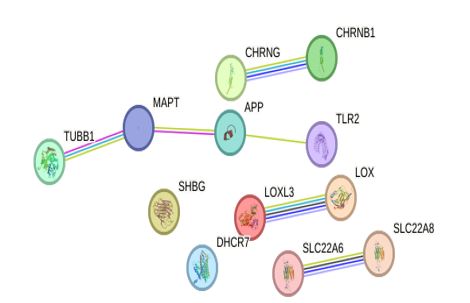
Figure 14: Protein- Protein interaction of Bauhinia Purpurea molecular Targets