Clinical Research and Clinical Case Reports
OPEN ACCESS | Volume 7 - Issue 1 - 2026
ISSN No: 2836-2667 | Journal DOI: 10.61148/2836-2667/CRCCR
Ben Jmaà Hèla1,3*, Mejri Samar1,3, Gueldich Majdi1,3, Ben Jmaà Tarak2,3, Hchaichi Nawel1,3, Dammak Aiman1,3, Ben Jmaà Mounir2,3, Frikha Imed1,3
1Department of cardiovascular and thoracic surgery Habib Bourguiba Hospital Sfax Tunisia.
2Department of infectious diseases Hedi Chaker Hospital Sfax Tunisia.
3Faculty of medicine of Sfax Tunisia.
*Corresponding authors: Ben Jmaà Hèla, Department of cardiovascular and thoracic surgery Habib Bourguiba Hospital Sfax Tunisia.
Received: November 08, 2021
Accepted: November 16, 2021
Published: November 18, 2021
Citation: Ben Jmaà Hèla, Mejri Samar, Gueldich Majdi, Ben Jmaà Tarak, Hchaichi Nawel et al (2021) “ Management of limb ischemia in patients with COVID-19 infection: A Tunisian center experience”. Clinical Research and Clinical Case Reports, 2(3); DOI: http;//doi.org/04.2021/1.1041
Copyright: © 2021 Ben Jmaà Hèla. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The novel coronavirus is characterized by multi-systemic involvement, and mortality is attributed mainly to the respiratory system involvement.
It is also associated with coagulopathy and both venous and arterial thromboembolic events that may lead to amputation of the affected limbs.
The management of limb ischemia is still a challenging issue in patients with Covid-19 infection.
This article describes our experience in managing limb ischemia by arterial occlusion in patients with COVID-19 infection.
Introduction:
An increasing number of the novel coronavirus-19 (COVID-19) infected patients may be asymptomatic or have mild, non-specific or atypical symptoms. Some of them may already have an ongoing, severe infection.
COVID 19 has demonstrated systemic effects, including significant immunologic, pulmonary, gastrointestinal, cardiac, and neurologic manifestations [1].
This disease induces hypercoagulability leading to the development of arterial and venous thrombosis; the underlying mechanism of which is still unclear.
We report our experience in the management of limb ischemia in patients with COVID-19 infection.
Material and methods:
A retrospective study was conducted in our institution on all patients with peripheral vascular involvement of COVID-19 infection.
Demographics, cardiovascular risk factors, clinical data, anatomic vascular occlusion, revascularization intervention method, COVID-19 testing, and results were collected.
Results:
Our series contains 12 patients with peripheral vascular involvement, and 2 patients with abdominal aorta thrombosis associated. Two patients suffered from ischemia of the upper limb, and the other patients suffered from ischemia of the lower limbs.
All of the patients were admitted to the COVID department for shortness of breath. The SARS-CoV 2 PCR test was positive in all patients.
Cardiovascular risk factors were present in 7 patients.
All of the patients received prophylactic anticoagulation, with a single dose daily enoxaparin.
Six patients had a severe ischemic syndrome with intense pain, anesthesia and paralysis of the extremities, pallor of the limbs which were cold to touch with absent distal pulses, and a cyanosis of the skin below the knee (figures 1 and 2).
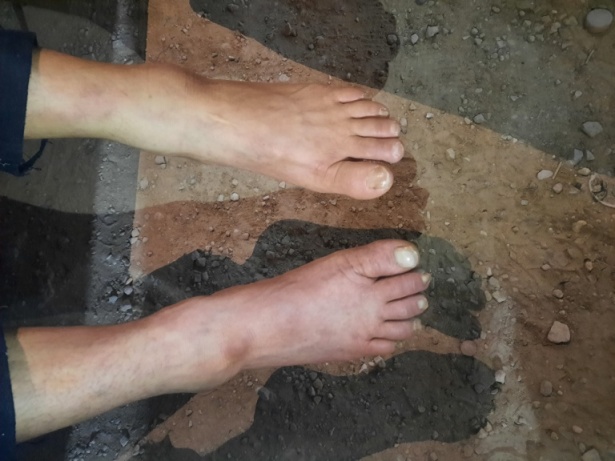
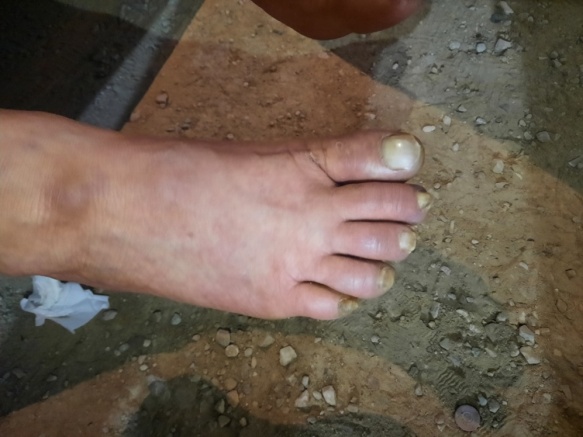
Figures 1 and 2: Photos showing cyanosis of the right leg in a patient suffering from limb ischemia (patient 7).
Four patients had a moderate ischemia with a mild pain, and without neurologic signs.
Three patients had an exceeded ischemia with gangrena.
One patient had an edema of the lower limb without coldness and pallor.
Clinical signs appeared during hospitalization for COVID infection in 9 cases, and after recovery from COVID infection in 5 patients.
Radiological investigation by doppler ultrasound was done 2 in patients. It revealed both arterial and venous ilio-femoral thrombosis in the patient with lower limb edema, and the absence of blood flow in the superficial femoral artery and the popliteal artery in 1 patient.
A CT scan was performed to assess the aorta and the large vessels in 7 patients, and it showed peripheral vascular occlusion in all these patients, associated with abdominal aortic floating thrombus in 2 patients (figures 3, 4, and 5).
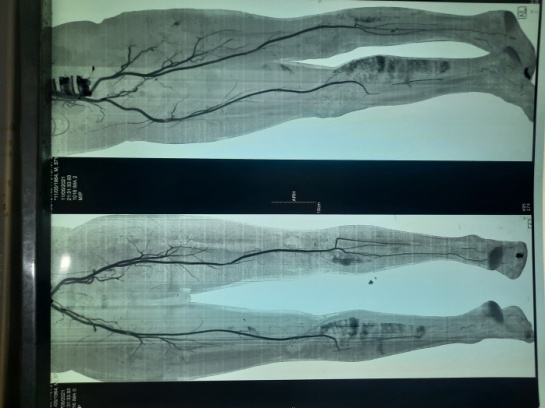
Figure 3: CT scan of 56-year-old man showing staged stenosis of the distal arteries (patient 7).
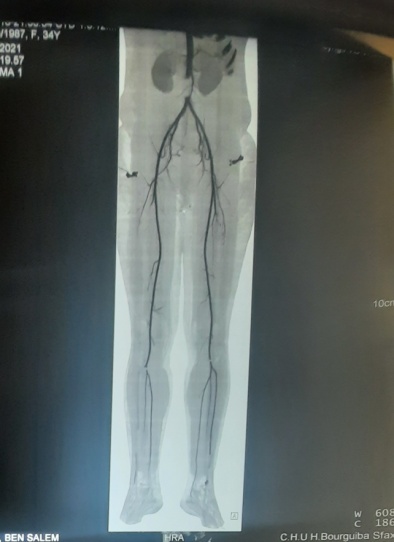
Figure 4: CT scan showing a thrombus of the abdominal aorta of a 35-year-old woman (arrow) (patient 10).
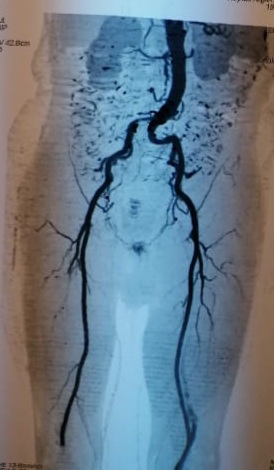
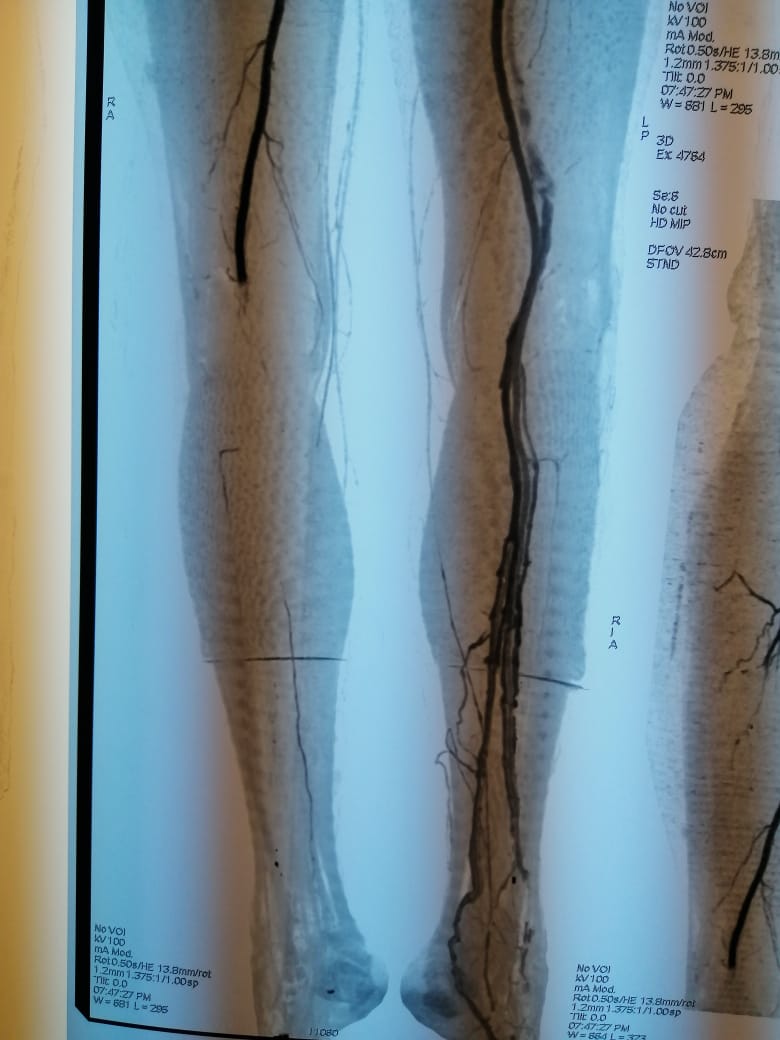
Figures 5 and 6: CT scan of 85-year-old man showing primary iliac artery thrombosis reducing the arterial lumen by 50 percent and embolic occlusion of the popliteal artery (arrows) (patient 11).
Electrocardiogram revealed normal sinus rhythm in 13 patients.
Echocardiographic study did not show any valvular pathology, endocarditis, or a patent foramen ovale in all of the patients.
Urgent thrombectomy was performed with Fogarty catheters in 6 patients. Abundant thrombi were removed proximally and distally, restoring arterial flow. Heparin was continued after the operation.
Medical treatment by intravenous heparin, acetyl salicylic acid and soludexide, without thrombectomy was performed in 5 patients because of the absence of severity of the ischemic signs which were moderate and not acute.
A limb amputation without revascularization was performed in 3 patients with exceeded ischemia.
The evolution of the patients after treatment was successful in 6 patients. Re-embolectomy was performed in 04 patients, and followed by continuous intravenous infusion of heparin. One patient died 2 days after the thrombectomy by respiratory distress.
The clinical, radiological, and therapeutic data of the patients are resumed in the table below:
|
Patient |
Age |
Gender |
Cardio-vascular Risk factors |
Ischemic signs appearance time compared to COVID infection |
The affected limb |
Clinical classification and diagnosis delay (hours) |
Exploration and affected artery |
Treatment |
Evolution of the ischemia |
|
1 |
70 |
Male |
none |
12 days later |
Left lower Limb |
Acute ischemia (2b)
72 hours |
Doppler ultrasound : No blood flow in SFA and PA |
Thrombectomy |
Immediate success Rethrombosis and amputation |
|
2 |
73 |
Male |
Diabetes |
9 days later |
Right lower limb |
Acute ischemia (2b)
72 hours |
CT scan : occlusion of the SFA |
Thrombectomy |
Immediate success Rethrombosis and amputation |
|
3 |
84 |
Female |
Diabetes |
60 days later |
Left upper limb |
Acute ischemia (2a)
08 hours |
none |
Thrombectomy |
Success |
|
4 |
50 |
Male |
tobacco |
09 days later |
Right lower limb |
Acute ischemia (2b)
09 hours |
none |
Thrombectomy |
*Immediate success *Rethrombosis Second thrombetcomy then amputation |
|
5 |
70 |
Male |
none |
10 days later |
Right lower limb |
Acute ischemia (2b)
48 hours |
CT scan : External Iliac artery thrombosis |
Thrombectomy |
Immediate success Rethrombosis then amputation |
|
6 |
82 |
Male |
HBP |
30 days later |
Left lower limb |
Moderate ischemia
72 hours |
CT scan : embolic occlusion of the lower 1/3 of SFA and PA |
Medical treatment |
Success : clinical improvement |
|
7 |
56 |
Male |
tobacco |
Same day |
Right lower limb |
Moderate ischemia
20 days |
CT scan : occlusion of PTA , ATA and PEA ( figures 1, 2 and 3) |
Medical treatment |
Success : clinical improvement |
|
8 |
82 |
Female |
HBP |
07 days later |
Left upper limb |
Exceeded ischemia
05 days |
none |
Amputation |
|
|
9 |
80 |
Male |
none |
07 days before COVID-19 diagnosis |
Right lower Limb |
Exceeded ischemia
07 days |
none |
Amputation |
|
|
10 |
34 |
Female |
none |
25 days later |
2 lower limbs |
Arteriopathy obliterans
07 days |
CT scan : *floating abdominal aortic thrombus * diffuse atheromatous infiltration * staged thrombosis of distal arteries in the two limbs ( figure 4) |
Medical treatement |
Success : clinical improvement |
|
11 |
85 |
Male |
none |
09 days later |
Right lower limb |
Acute ischemia (2a)
09 hours
|
CT scan : *Floating thrombus of the abdominal aorta *primary iliac artery thrombosis reducing the arterial lumen by 50 percent *embolic occlusion of PA (figures 5 and 6) |
thrombectomy |
Death of the patient two days after the thrombectomy |
|
12 |
85 |
Male |
HBP |
20 days later |
Left lower limb |
Exceeded ischemia
05 days |
none |
Amputatin |
|
|
13 |
56 |
Male |
none |
10 days later |
Right lower limb |
Moderate ischemia (1) + Edema |
Doppler ultrasound: *Arterial and venous ilio-femoral thrombosis
|
Medical treatment |
Success : clinical improvement |
|
14 |
60 |
Male |
none |
25 days later |
Right lower limb |
Moderate ischemia |
CT scan: Staged thrombosis of PEA and PTA |
Medical treatment |
Success : clinical improvement |
Table: Clinical, radiological, and therapeutic data of the patients:
Abbreviations:
HBP: high blood pressure
PA: Popliteal artery
SFA: superficial femoral artery
ATA: anterior tibial artery
PTA: posterior tibial artery
PEA: Peroneal artery
Discussion:
The novel coronavirus disease-2019 (COVID-19) renders thrombotic complications in both arterial and venous supply. The pro-inflammatory cytokines that increase over the course of infection activate the coagulation cascade by multiple mechanisms, including increased expression of tissue factor and endothelial activation [2]. That’s why patients with novel coronavirus can suffer from thrombosis in the absence of traditional risk factors for acute thromboembolism [3].
These thrombotic events have been reported in up to 4.5% of relatively healthy individuals with COVID-19 in the patients not requiring hospitalization [4]. Some cases were reported after discharge from COVID pneumonia, and they worsen the prognosis of the patients [5].
Surgeons continue to see a steady stream of threatening limb ischemic cases that require emergent surgical care.
Some clinical cases and studies were reported in the literature, especially increased involvement of multi-level disease and more likely to require amputations.
Khan A et al [6] described a case of multi-focal ischemic stroke and acute limb ischemia in a 63-year-old man hospitalized SARS CoV2 infection managed medically.
Lodigiani et al [7] reported a rate of 27.6% for thrombotic events in intensive care unit patients, and a rate of 6.6% for general ward patients.
Intraluminal floating thrombus in aorta was also observed in many ischemic events associated with COVID-19 patients, such as the case of 2 of our patients [8, 9].
Other vascular locations may be involved.
Ghaitha Al Mahruqi et al [10] presented 2 cases of acute mesenteric ischemia in COVID positive patients.
In a study that included 3334 individuals (829 intensive care unit patients and 2505 non-intensive care unit patients), Bilalogluet al. reported stroke in 1.6% and myocardial infarction in 8.9% [11].
The risk of thromboembolic complications continues even after recovery from COVID infection and discharge from hospital [5].
Bastopcu M [5] reported a case of an extensive arterial thrombus in the left leg of a Covid-19 pneumonia.
Borrelli M.P et al [12] reported 3 cases that had different delayed vascular arterial complications, which occurred after the index infection, with a negative nasopharyngeal swab and Covid-19 systemic symptoms resumption.
Arterial involvement after recovery from COVID was observed in 5 of our patients (patients 3, 6, 10, 12, and 14)
Conservative therapies such as heparin, vasodilator drugs and fibrinolytic agents are only adjunct to embolectomy which is the method of choice and applicable in almost all cases [8, 13].
The principles in surgical managing these COVID positive patients are to ensure the safety of the medical team at all times, prioritize life of the limb, do the minimum necessary limb-saving procedure, minimize repeated operations during the contagious period, and defer all other procedures until the patient has recovered from the acute viral infection [14].
A multidisciplinary approach involving the patient, surgeon, anesthesiologist, and intensivist must be taken to ensure the risks are justified [14].
The current guideline for anesthesia in COVID positive patients is to minimize aerosolization [15]. Fortunately, many limb salvage procedures can be performed under local or regional anesthesia augmented with sedation, obviating the need for general anesthesia. All of our patients who underwent surgery are operated under local anesthesia.
In the postoperative period, a COVID-19 positive patient who has undergone a major procedure should be nursed in intensive care unit [16]. Anticoagulant therapy must be continued.
Medical treatment without embolectomy is sometimes indicated when the ischemic signs are moderate, not acute, and anatomic lesions are distal. That was the therapeutic mean used in 5 of our patients (patients 6, 7, 10, 13, and 14), with favourable evolution.
The rate of limb loss by rethrombosis in the postoperative period is high because of the hypercoagulable state of the patients with Covid-19 [8]. Also, a damaged arterial intima, the excessive adherence of the thrombi to the intima or an occluded distal arterial tree may be accused of the re-occlusion after revascularization procedures.
Also, morbidity and mortality in this subset of patients is likely to be high [10].
In our series, only one patient died 2 days after revascularization by respiratory distress, and the rate of limb loss was high: patients underwent amputation without revascularization because the ischemia was exceeded with gangrena (patients 8, 9, and 12), and 4 patients underwent thrombectomy followed by rethrombosis and then amputation (patients 1, 2, 4, and 5).
Conclusion:
Covid-19 associated coagulopathy can cause thrombosis in the arterial circulation with poor outcomes.
These cases contribute to the literature on the systemic thromboembolic complications associated with COVID-19.
The clinical management and outcomes in the arterial revascularization procedures of the patients with vascular complications, is still a challenging issue in COVID-19 infection associated with limb ischemia.
So, it is highly recommended to include antithrombotic treatment in every hospitalized COVID-19 patient, because of the possibility of venous and arterial hypercoagulability states, which frequently complicate patients with COVID-19 infection.